
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 5.35SP
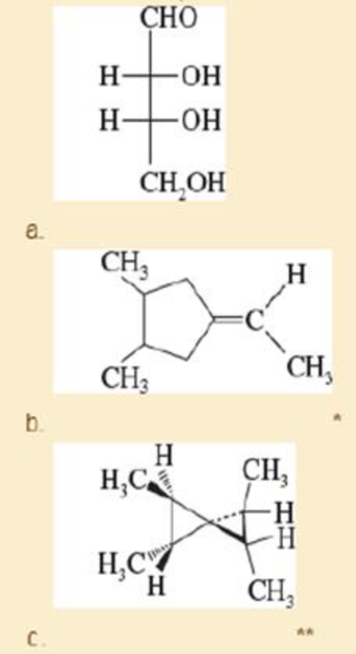
For each structure,
- 1. draw all the stereoisomers.
- 2. label each structure as chiral or achiral.
- 3. give the relationships between the stereoisomers (enantiomers, diastereomers).
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Label the chiral center ( R or S) , then draw the structures enantiomer.
The structure of 4 isomers of an aldotetrose carbohydrate are given.
1) select every structure that is a diastereomer of structure D
A, B, or C?
2) select every structure that is a enantiomer of structure C
D, B, or A?
3) select every structure that is a stereoisomer of structure D
A, B, or C
1. Draw all of the stereoisomers of 2,4-dichloropentane. Indicate the relationship between these stereoisomers, such as enantiomers, diastereomers, or meso compounds.
Chapter 5 Solutions
Organic Chemistry (9th Edition)
Ch. 5.2 - Determine whether the following objects are chiral...Ch. 5.2A - Prob. 5.2PCh. 5.2B - Prob. 5.3PCh. 5.2B - Prob. 5.4PCh. 5.2C - Prob. 5.5PCh. 5.3 - Prob. 5.6PCh. 5.3 - Prob. 5.7PCh. 5.4D - Prob. 5.8PCh. 5.4D - Prob. 5.9PCh. 5.4D - Prob. 5.10P
Ch. 5.5 - Prob. 5.11PCh. 5.7 - When optically pure (R)-2-bromobutane is heated...Ch. 5.7 - Prob. 5.13PCh. 5.8 - Prob. 5.14PCh. 5.9B - Draw three-dimensional representations of the...Ch. 5.10A - For each sot of examples, make a model of the...Ch. 5.10A - Draw a Fischer projection for each compound....Ch. 5.10B - Prob. 5.18PCh. 5.10C - For each Fischer projection, label each asymmetric...Ch. 5.11C - Prob. 5.20PCh. 5.13 - Prob. 5.21PCh. 5.13 - Prob. 5.22PCh. 5.15 - Prob. 5.23PCh. 5.16A - Prob. 5.24PCh. 5 - The following four structures are naturally...Ch. 5 - For each structure, 1. star () any asymmetric...Ch. 5 - Prob. 5.27SPCh. 5 - Prob. 5.28SPCh. 5 - Prob. 5.29SPCh. 5 - Prob. 5.30SPCh. 5 - Prob. 5.31SPCh. 5 - Prob. 5.32SPCh. 5 - Prob. 5.33SPCh. 5 - Prob. 5.34SPCh. 5 - For each structure, 1. draw all the stereoisomers....Ch. 5 - Prob. 5.36SPCh. 5 - Prob. 5.37SPCh. 5 - 3,4-Dimethylpent-1-ene has the formula...Ch. 5 - A graduate student was studying enzymatic...Ch. 5 - Prob. 5.40SPCh. 5 - Prob. 5.41SP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Fully developed conditions are known to exist for water flowing through a 25-nim-diameer tube at 0.01 kg/s and ...
Fundamentals of Heat and Mass Transfer
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
16.43 The following pictures represent solutions at various stages in thetitration of a weak diprotic acid with...
Chemistry (7th Edition)
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living by Chemistry
What is the pH range for acidic solutions? For basic solutions?
Introduction to Chemistry
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Label each molecule with chiral centers (R) and (S). Which ones have more than one?arrow_forward1a. How many stereogenic centers are present 1c. Draw a three-dimensional structure of a in the structure below? Indicate them with asterisk(s). How many stereoisomers stereoisomers are possible? chiral compound with the molecular formula of C4H4Cl₂ that does not have a stereogenic carbon. In addition, draw the enantiomer of this compound. Number of stereogenic centers: Number of stereoisomers possible: 1b. Draw one of the two most stable stereoisomers of the compound in 1a using a planar structure with wedges and dashes. Now draw it in its preferred chair conformation. 1d. Draw two meso compounds with the molecular formula of C7H14.arrow_forward2. Mark any chirality centers (asymmetric carbons) with a *. How many stereoisomers are possible? Brarrow_forward
- ball & stick + labels stereoisomer(s) e. Are the two molecules enantiomers? yes no ball & stick a. How many chiral carbons are there in the molecule on the left? chiral carbon(s) b. What is the total number of stereoisomers in the group that includes the molecule on the left? stereoisomer(s) c. How many chiral carbons are there in the molecule on the right? chiral carbon(s) d. What is the total number of stereoisomers in the group that includes the molecule on the right? + labels Previous Next Save and Exitarrow_forwardBecause there is usually slow interconversion between the two isomeric forms at room temperature. Because there is usually rapid interconversion between the two isomeric forms at room temperature. Because chirality only exists with the tetrahedral carbon atoms. Because four bonds a are needed to define a stereogenic center.arrow_forward(a) assign R or S configuration to each chiral center, (b) Which compound are enantiomers? (c) Which compounds are diastereomers?arrow_forward
- 15. Naming Stereoisomers with Two Chiral Carbons Using the RS System The (RR) isomer of methyphenidate (Ritalin) is used to treat attention deficit hyperactivity disorder (ADHD). The (S.S) isomer is an antidepressant. Identify the two chiral carbons in the structure below. Is this the (R.R) or the (S.S) isomer? Draw the other isomer. HN- H. ..arrow_forwardFor each pair of compounds, please explain if they are identical. comstitutional isomers, enantiomers, or diastereomers.arrow_forwardIdentify the relationship between the two structures. a. Identical b. Enantiomers c. Diastereomers d. Constitutional isomers The molecule to the right has chiral carbon atoms and can have a total of stereoisomers. а. one; two b. two; four c. three; eight d. four; eight с.arrow_forward
- A. Give your comment(s) on the following questions below: 1. Are all chiral molecules enantiomers? Why or why not? 2. What is the difference between enantiomers and diastiomers ? 3. What are the differences between stereoisomers, structural isomers and constitutional isomers?arrow_forwardConsider the compound below. a) Draw the structure showing stereochemistry, in which carbon 1 has S configuration and carbon 2 has R configuration. b) Draw the structure showing stereochemistry, in which carbons 1 and 2 have S configuration. c) are the two structures from part a and b diastereomers, identical, enantiomers, or unrelated?arrow_forward5. Assign R and S to each chiral center. Br H. CI NH2 H, OH H... OCH3 CH3 H. NC A A [ [ Choose ] [ Choose ] [ Choose ] C [ [ Choose ] Darrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License