
Concept explainers
(a)
Interpretation: The given groups are to be ranked in the order of decreasing priority.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The priorities of the atoms that are directly attached to the stereogenic center are assigned in the order of decreasing
Answer to Problem 5.45P
The decreasing order of priority is
Explanation of Solution
The priority can be assigned by numbering the groups which are bonded to the chiral carbon atom based on the molecular weight and electronegativity.
In the given groups, the atomic number of nitrogen is
The decreasing order of priority for the given groups is
(b)
Interpretation: The given groups are to be ranked in the order of decreasing priority.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The priorities of the atoms that are directly attached to the stereogenic center are assigned in the order of decreasing atomic number. An atom of highest atomic number gets highest priority.
Answer to Problem 5.45P
The decreasing order of priority for the given groups is
Explanation of Solution
The priority can be assigned by numbering the groups which are bonded to the chiral carbon atom based on the molecular weight and electronegativity.
The given groups are
Therefore, the decreasing order of priority for the given groups is
The decreasing order of priority for the given groups is
(c)
Interpretation: The given groups are to be ranked in the order of decreasing priority.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The priorities of the atoms that are directly attached to the stereogenic center are assigned in the order of decreasing atomic number. An atom of highest atomic number gets highest priority.
Answer to Problem 5.45P
The decreasing order of priority is
Explanation of Solution
The priority can be assigned by numbering the groups which are bonded to the chiral carbon atom based on the molecular weight and electronegativity.
In the given groups, the atomic number of nitrogen is
Hence, the decreasing order of priority is
The decreasing order of priority for the given groups is
(d)
Interpretation: The given groups are to be ranked in the order of decreasing priority.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The priorities of the atoms that are directly attached to the stereogenic center are assigned in the order of decreasing atomic number. An atom of highest atomic number gets highest priority.
Answer to Problem 5.45P
The decreasing order of priority is
Explanation of Solution
The priority can be assigned by numbering the groups which are bonded to the chiral carbon atom based on the molecular weight and electronegativity.
In the given groups, the atomic number of carbon is
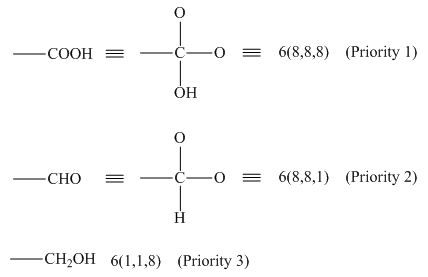
Figure 1
Hence, the decreasing order of priority is
The decreasing order of priority for the given groups is
(e)
Interpretation: The given groups are to be ranked in the order of decreasing priority.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The priorities of the atoms that are directly attached to the stereogenic center are assigned in the order of decreasing atomic number. An atom of highest atomic number gets highest priority.
Answer to Problem 5.45P
The decreasing order of priority is
Explanation of Solution
The priority can be assigned by numbering the groups which are bonded to the chiral carbon atom based on the molecular weight and electronegativity.
In the given groups, the atomic number of chlorine is
The decreasing order of priority for the given groups is
(f)
Interpretation: The given groups are to be ranked in the order of decreasing priority.
Concept introduction: A carbon atom that has four nonequivalent atoms or groups attached to it is known as chiral carbon atom. Chiral carbon centers are also called as asymmetric or stereogenic centers.
The priorities of the atoms that are directly attached to the stereogenic center are assigned in the order of decreasing atomic number. An atom of highest atomic number gets highest priority.
Answer to Problem 5.45P
The decreasing order of priority for the given groups is
Explanation of Solution
The priority can be assigned by numbering the groups which are bonded to the chiral carbon atom based on the molecular weight and electronegativity.
An atom of highest atomic number gets highest priority. Therefore, in the given groups,
For an atom which is a part of multiple bonds, the priority is assigned by treating the number of multiple bonded atoms equivalent to the number of singly bonded atoms. Among,
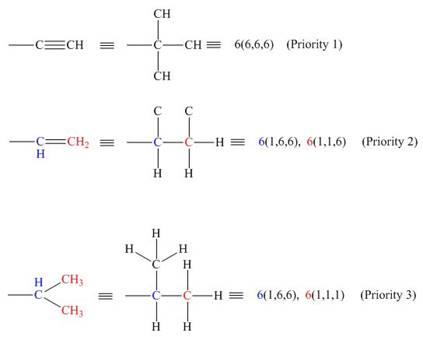
Figure 2
Hence, the decreasing order of priority for the given groups is
The decreasing order of priority for the given groups is
Want to see more full solutions like this?
Chapter 5 Solutions
Organic Chemistry
- Choose the correct product for the given reaction. CF3 HBr ? CF3 CF3 Br A. Br Br CF3 Br CF3 D. Br С. Br A B B. ооо оarrow_forwardTo preview the image click here For the following two structures compared in parts A and B, determine whether they are enantiomers OR diastereomers OR the same structure drawn differently (non-bonding electrons not included for clarity). A. B. HO H Н. H3C CH3 H HO Me CI HO H H3C CH3 "OH H Me CL ape-oroflocktem isarrow_forwardDetermine which reactions would occur (substitution, elimination, both, or neither). CH;CH,OH a. SN1 O b. E1 C. SN1 & E1 O d. SN2 е. Е2 O f. SN2 & E2 g. No reactionarrow_forward
- Which group in each pair is assigned the higher priority in R,S nomenclature? а. — CDз, — СH b. - CH(CH3)2, - CH2OH c. - CH,CI, - CH,CH;CH,Br d. - CH,NH2, - NHCH,arrow_forwardPART 2 FOR EACH REACTION THAT FOLLOWS: 1. Name the functional groups & reactants 2. Name the reaction 3. Draw the major product K. L. M. N. P. Nall THE Q. 1. Hg(0Ac), MeOH 2. NaBH₂ O. m HO 1.9-BBN 2. NaOH, H₂O₂ OH OMe -OH Na Cr₂O H₂SO₂, H₂O PCC DCM PCC DCM IM NaOH(aq). (H₂C),CO cat. TsOHarrow_forwardWhich group in each pair is assigned the higher priority in R,S nomenclature? a.−CD3, −CH3 b.−CH(CH3)2, −CH2OH c.−CH2Cl, −CH2CH2CH2Br d.−CH2NH2, −NHCH3arrow_forward
- Which are secondary alcohols? H. CH; CH; c-c-OH H,C H H;C-C CH; H `CHCH, OH II III IV O A. I, II B. II, II C. III, IV O D. I, IVarrow_forwardH3C- -Br + а. b. H3C CH3 2 CH3CH,NH2arrow_forwardA. Classify each alkyl halide as 1°, 2°, or 3º. 1. CH;CH2CH2CH2CH2-Br Br F 2. 5. CH3 CH3CH2-C-CH½CH3 CI CH3 CH3-C-CHCH3 3. 6. CH3 ČI CH3 CH;CH2-C-CH2I ČH3 7.arrow_forward
- 9. Part 1: Draw the structure for compound A. Part 2: Draw the structure for compound B. Part 3: Identify the required reagent(s) for step 1. Select the single best answer. A) NaOH, H2O B) NaNH2, NH3 C) H2SO4 D) NH3 Part 4:Identify the required reagents for step 3. Select the single best answer. A) Na, NH3 B) H2, Pd C) H2, Lindlar catalyst D) NaNH2, NH3arrow_forward4. Provide the reagent(s) needed to accomplish the following transformations. Some conversions may require more than one step. If more than one step is required, be sure to enumerate each step. a. H. b. Ph. HO, c. HO, d. HO, OH е. OMe OMe Ph. OMe Ph ОН f. OH NH NH2 OMe OMearrow_forwardOrder the following in increasing priority A. -C, -CH, -OL B. -CH3, -CH2OH, -CH2CH3 C. -C≡CH, -CH゠CH2, -CH゠Oarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





