
Biology 2e
2nd Edition
ISBN: 9781947172517
Author: Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher: OpenStax
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7, Problem 2VCQ
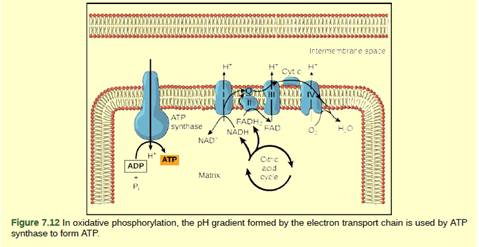
Figure 7.12 Cyanide inhibits cytochrome c oxidase, a component of the electron transport chain. If cyanide poisoning occurs, would you expect the pH of the intermembrane space to increase or
decrease? What effect would cyanide have on ATP synthesis?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Cyanide inhibits cytochrome c oxidase, a component of the electron transport chain. If cyanide poisoning occurs, would you expect the pH of the intermembrane space to increase or decrease? What effect would cyanide have on ATP synthesis?
Figure 7.11 Dinitrophenol (DNP) is an "uncoupler"
that makes the inner mitochondrial membrane
"leaky" to protons. It was used until 1938 as a weight-
loss drug. What effect would you expect DNP to have
on the change in pH across the inner mitochondrial
membrane? Why do you think this might be an
effective weight-loss drug?
Intermembrane
space
Mitochondrial
matrix
ATP Synthase
ADP
Inner mitochondrial
membrane
ATP
Figure 7.11 ATP synthase is a complex, molecular machine that uses a proton (H) gradient to form ATP from
ADP and inorganic phosphate (Pi). (Credit: modification of work by Klaus Hoffmeier)
As electrons move from Complex I & II to Complex IV, H+ ions accumulate in the intermembrane space. Describe how these ions are then used to generate ATP using the following terms: ATP synthase, ADP, ATP, electrochemical gradient, chemiosmosis, intermembrane space, matrix.
Chapter 7 Solutions
Biology 2e
Ch. 7 - Figure 7.11 Dinitrophenol (DNP) is an "uncoupler"...Ch. 7 - Figure 7.12 Cyanide inhibits cytochrome c oxidase,...Ch. 7 - (Figure 7.14) Tremetol, a metabolic poison found...Ch. 7 - The energy currency used by cells is ATP ADP AMP...Ch. 7 - A reducing chemical reaction. reduces the compound...Ch. 7 - During the second half of glycolysis, what occurs?...Ch. 7 - What is removed from pyruvate during its...Ch. 7 - What do the electrons added to NAD+ do? They...Ch. 7 - GTP or ATP is produced during the conversion of...Ch. 7 - How many NADU molecules are produced on each turn...
Ch. 7 - What compound receives elections from NADH? FMN...Ch. 7 - Chemiosmosis involves. the movement of electrons...Ch. 7 - Which of the following fermentation methods can...Ch. 7 - A major connection for sugars in glycolysis is...Ch. 7 - Beta-oxidation is. the breakdown of sugars the...Ch. 7 - The effect of high levels of ADP is to__inv __...Ch. 7 - The control of which enzyme exerts the most...Ch. 7 - Why is it beneficial for cells to use ATP rather...Ch. 7 - Nearly all organisms on Earth carry out some form...Ch. 7 - Because they lose their mitochondria during...Ch. 7 - What is the primary difference between a circular...Ch. 7 - How do the roles of ubiquinone and cytochrome c...Ch. 7 - What accounts for the different number of ATP...Ch. 7 - What is the primary difference between...Ch. 7 - Would you describe metabolic pathways as...Ch. 7 - How does citrate from the citric acid cycle affect...Ch. 7 - Why might negative feedback mechanisms be more...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Which of the following is a vector: a person's height, the altitude on Mt. Everest, the age of the Earth, the b...
College Physics
Consider two hypothetical recessive autosomal genes a and b, where a heterozygote is testcrossed to a double-ho...
Concepts of Genetics (12th Edition)
3. CAUTION Why is genetic drift aptly named?
a. It causes allele frequencies to drift up or down randomly.
b. I...
Biological Science (6th Edition)
Match the following examples of mutagens. Column A Column B ___a. A mutagen that is incorporated into DNA in pl...
Microbiology: An Introduction
4. What five specific threats to biodiversity are described in this chapter? Provide an example of each.
Biology: Life on Earth
41. Humans vary in many ways from one another. Among many minor phenotypic differences are the following five i...
Genetic Analysis: An Integrated Approach (3rd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Which of these molecules associated with electron transport is in the reduced state? Choose from the following: (A) cytochrome a3+++ (b) cytochrome a3++arrow_forwardYou are studying the process of oxidative phosphorylation in the lab. You isolate several mitochondria from cells and place them in a buffered solution with a low pH and observe that the mitochondria begin to synthesize ATP. Which of the following is the best explanation for this observation? F It increases the conc. of OH-, causing the mitochondria to pump H+ to the intermembrane space. It increases the diffusion of H+ from the mitochondrial matrix to the intermembrane space. It increases the diffusion of H+ from the intermembrane space to the matrix. It increases the conc. of OH- in the mitochondrial matrix.arrow_forwardPlease answer this question in detail because I don't understand it. Thank you. Which of the following best describes the role of chemiosmosis in oxidative phosphorylation? (A) Chemiosmosis is the movement of electrons down their electrochemical gradient, which generates energy in the electron transport chain. (B) Chemiosmosis is the chemical breakdown of ATP into ADP and inorganic phosphate. (C) Chemiosmosis is the movement of chemicals dissolved in water down their concentration gradient, across the mitochondrial membrane. (D) Chemiosmosis is the chemical breakdown of water into hydrogen and oxygen ions at the final stage of the electron transport chain. (E) Chemiosmosis is the movement of ions down their electrochemical gradient, which generates ATP.arrow_forward
- The graph shows how the free‑energy change for the hydrolysis of ATP varies as a function of Mg2+ concentration. Note that pMg = –log[Mg2+]. What does the graph tell you about the relationship between Mg2+ concentration and ATP hydrolysis? What is the mechanism that explains the relationship between Mg2+ concentration and ATP hydrolysis?arrow_forwardThe mitochondrion pumps H+ from the matrix into the intermembrane space. Which region is more acidic, the matrix or the intermembrane space? Why?arrow_forwardEach proton that moves across the membrane releases about 14 kJ/mol of energy. Given that ATP requires 30.5 kJ/mol of energy to form, how many protons cross the membrane per ATP synthesized? (Hint: can you have half a proton?)arrow_forward
- Although the outer mitochondrial membrane is permeable to all small molecules, the inner mitochondrial membrane is essentially impermeable in the absence of specific transport proteins. Consider this information answer: The ATP generated by oxidative respiration is used throughout the cell. The majority of ATP production occurs in the mitochondrial matrix. How do you think ATP is made accessible to enzymes in the cytosol and other organelles?arrow_forwardChoose the correct answer: 1- How does ETC lead to ATP synthesis: A) Hydrogen ions diffuse from the intermembrane space to the matrix through ATP synthase.B) Hydrogens jump from the inner membrane to the outer membrane and out through ATP synthase.C) Hydrogens diffuse from the intermembrane space to the outer membrane through ATPase.D) Protons are pumped from the matrix to the intermembrane space through ATP synthase. 2- TCA cycle occurs in aerobic respiration because : A) Oxygen has a catalytic functionB) Oxygen is a reactantC) Electron transport chain requires aerobic conditions to operate. D) All of the above.arrow_forwardThermogenin in the inner membrane of brown fat cell mitochondria does all of the following except A) decrease the proton gradient. B) uncouple ATP synthesis from electron transport. C) generate heat. D) decrease ATP synthesis. E) increase ATP synthesisarrow_forward
- The production of a hydrogen ion concentration gradient powers the production of ATP in both photosynthesis and cellular respiration. 2,4-Dinitrophenol (DNP) is an organic compound that 'undoes' the hydrogen ion concentration gradient without the production of ATP. It does this by increasing the membrane permeability to hydrogen ions. One use of DNP is as a herbicide.For a brief period in the 1930s, DNP was marketed as a diet pill. Because DNP prevents the production of ATP, the human body will begin to use alternate forms of energy. The result is an increase in the metabolism of fats in the body, thus reducing total body fat. In the presence of DNP, the energy that normally would be converted to ATP for use in the cells is converted to heat instead, causing dangerously high body temperatures.DNP is classified as an illegal substance in Canada and the U.S. although it is still marketed as a commercial chemical. In recent years, several deaths have been reported, primarily in the…arrow_forwardWhat would be the effect on ATP production during chemiosmosis and oxidative phorphorylation, if the pH within the inner membrane space of the mitochondrion were decreased, assuming no effect of pH on the structure of proteins in the membrane? a) it would increase b) it would decrease c)it would stay the samearrow_forwardWhat are two methods that cells can use to allow glycolysis to continue to produce ATP. in anaerobic conditions? Describe these two methods including the products and reactants.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Concepts of Biology
Biology
ISBN:9781938168116
Author:Samantha Fowler, Rebecca Roush, James Wise
Publisher:OpenStax College
Anaerobic Respiration; Author: Bozeman Science;https://www.youtube.com/watch?v=cDC29iBxb3w;License: Standard YouTube License, CC-BY