
Chemistry In Context
9th Edition
ISBN: 9781259638145
Author: Fahlman, Bradley D., Purvis-roberts, Kathleen, Kirk, John S., Bentley, Anne K., Daubenmire, Patrick L., ELLIS, Jamie P., Mury, Michael T., American Chemical Society
Publisher: Mcgraw-hill Education,
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7.11, Problem 7.16YT
You Decide Could Metals Become Extinct?
In 2015, the American Chemical Society published this “Periodic Table of Endangered Elements”:
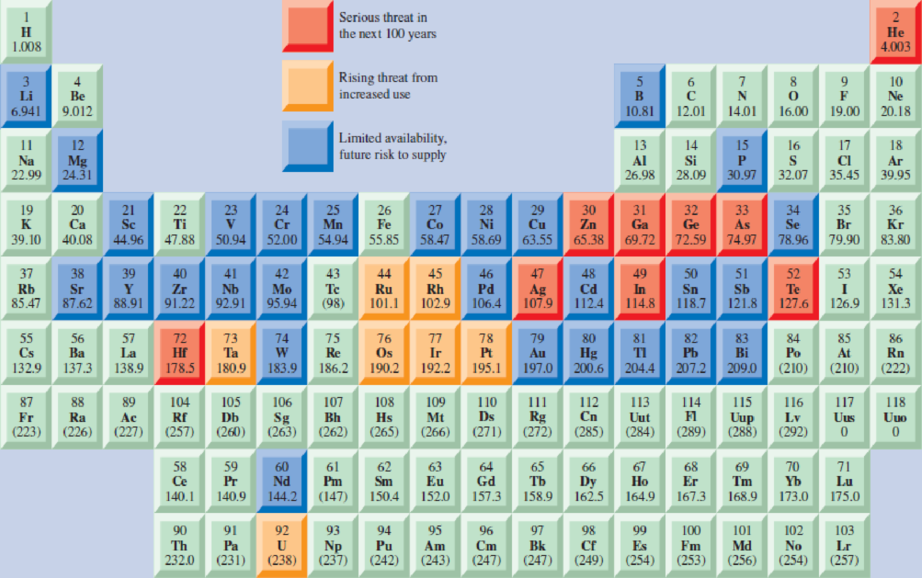
- a. Is it really possible for a metal to become “extinct”?
- b. Which of the highlighted elements above are currently in demand for energy storage devices (batteries, fuel cells, supercapacitors)?
- c. Are there any non-highlighted elements that are currently being used, or tested for future use, in energy-storage applications?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
5 Elements are -
1) Sodium.
2) potassium
3) calcium
4) magnesium
5)lithium
Rank them from 1 to 5, 1 being the most important and 5 being the least important. Explain how they are medically relevant
If informative, I will upvote, I promise pls pls
mHMAC3ccdb6aa11676716c83835709749d96e5#10001
Translate
Reading list
1 of 11
I Review | Constants Periodic Table
Part A
Julia spent $50 on a book. The cost of the book was $18 less than twice what she spent on a
backpack. How much did the backpack cost?
Express your answer numerically in dollars.
• View Available Hint(s)
Hint 1. Identify the mathematical equation
Hint 2. Identify the expression for the cost of the backpack
Ph
Ph
2. (Across) Heaviest element
that a star can fuse. (symbol)
3. (Across) Agua Pesada
(Formula)
1. (Down) It is a gain and lose situation.
Ph
4. (Across) In IUPAC naming
this is the prefix of a halogen.
1. (Across) Radioactive gas
that builds up inside buildings.
OH
2
1
3
5
5. (Across) Roentgen
#
Ph
First Name
Ph
Chapter 7 Solutions
Chemistry In Context
Ch. 7.1 - Prob. 7.2YTCh. 7.2 - Skill Building Coulombs The SI unit of electric...Ch. 7.2 - Prob. 7.4YTCh. 7.4 - Prob. 7.5YTCh. 7.4 - Scientific Practices The Shift from NiCd to NiMH...Ch. 7.5 - Scientific Practices The Battery in Your Car Lets...Ch. 7.7 - Prob. 7.9YTCh. 7.8 - Prob. 7.10YTCh. 7.9 - Prob. 7.11YTCh. 7.9 - Prob. 7.12YT
Ch. 7.10 - Prob. 7.13YTCh. 7.10 - Prob. 7.14YTCh. 7.11 - Skill Building Metal Refining (Smelting) The...Ch. 7.11 - You Decide Could Metals Become Extinct? In 2015,...Ch. 7.11 - Prob. 7.17YTCh. 7 - You Decide The Baghdad Battery Alessandro Volta is...Ch. 7 - Prob. 1QCh. 7 - Prob. 2QCh. 7 - Which chemical species gets oxidized and which...Ch. 7 - What is the difference between a galvanic cell and...Ch. 7 - Two common units associated with electricity are...Ch. 7 - Consider the galvanic cell pictured. A coating of...Ch. 7 - Prob. 7QCh. 7 - a. How does the voltage from a tiny AAA alkaline...Ch. 7 - Identify the type of galvanic cell commonly used...Ch. 7 - The mercury battery has been used extensively in...Ch. 7 - Prob. 12QCh. 7 - During the conversion of O2(g) to H2O(l) in a fuel...Ch. 7 - How does the reaction between hydrogen and oxygen...Ch. 7 - This diagram represents the hydrogen fuel cell...Ch. 7 - What is a PEM fuel cell? How does it differ from...Ch. 7 - How do PEM fuel cells allow H2 and O2 to combine...Ch. 7 - In addition to hydrogen, methane also has been...Ch. 7 - Prob. 19QCh. 7 - Potassium and lithium both are reactive Group 1...Ch. 7 - What challenges keep hydrogen fuel cells from...Ch. 7 - Explain the concept of energy density of a battery...Ch. 7 - Describe how a normal AA battery stores and...Ch. 7 - Prob. 24QCh. 7 - What is the difference between an electrolytic...Ch. 7 - Provide some differences between a leadacid...Ch. 7 - Describe the importance of a separator in primary...Ch. 7 - The company ZPower is promoting its silverzinc...Ch. 7 - Prob. 29QCh. 7 - Prob. 30QCh. 7 - Describe some advantages and disadvantages of...Ch. 7 - You never need to plug in Toyotas gasolinebattery...Ch. 7 - Prob. 33QCh. 7 - Hydrogen is considered an environmentally friendly...Ch. 7 - Fuel cells were invented in 1839 but never...Ch. 7 - Hydrogen and methane both can react with oxygen in...Ch. 7 - Engineers have developed a prototype fuel cell...Ch. 7 - Prob. 38QCh. 7 - Describe some similarities and differences between...Ch. 7 - Prob. 40QCh. 7 - Prob. 41QCh. 7 - Prob. 42QCh. 7 - Small quantities of hydrogen gas can be prepared...Ch. 7 - Prob. 44QCh. 7 - Although Alessandro Volta is credited with the...Ch. 7 - Prob. 46QCh. 7 - Prob. 47QCh. 7 - What is the tragedy of the commons? How does this...Ch. 7 - How can the principles of green chemistry be...Ch. 7 - If all of todays technology presently based on...Ch. 7 - Consider these three sources of light: a candle, a...Ch. 7 - Prob. 52Q
Additional Science Textbook Solutions
Find more solutions based on key concepts
16.43 The following pictures represent solutions at various stages in thetitration of a weak diprotic acid with...
Chemistry (7th Edition)
1. What did each of the following scientists contribute to our knowledge of the atom?
a. William Crookes
b. E...
Chemistry For Changing Times (14th Edition)
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living by Chemistry
Give one example from main group chemistry that illustrates each of the following descriptions: (a) Covalent ne...
General Chemistry: Atoms First
covered a synthesis of alkynes by a double dehydrohalogenation of dihalides. A student tried to convert trans-2...
Organic Chemistry (9th Edition)
Give the IUPAC name for each compound.
Organic Chemistry
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The vitamin niacin (nicotinic acid,C6H5NO2 ) can he isolated from a variety of natural sources, such as liver, yeast, milk, and whole grain. It also can be synthesized from commercially available materials. Which Source of nicotinic acid, from a nutritional view, is best for use in a multivitamin tablet? Why?arrow_forward1) Describe why "new" elements (man-made elements) are created, or why do we benefit in creating them? 2) Explain why creating new elements is difficult. 3) In general, how are man-made elements created (give 2 examples). 4) Discuss generally how they differ from "natural elements". And give 2 examples of man-made elements. 5) And discuss whether you believe humans can "create" naturally occurring elements if we have a scarcity of those elements on earth (either because we're depleting them from earth or if they are not abundant).arrow_forwardAfter reading the text in What's new. Let us see how far you have learned this topic. Activity 1.2: Fill in what is missing in the ZApen table. Use a separate sheet of paper for your answer. Element Symbol (p+) (e) (ne) A Ar 18 40 8 N 7 7 15 25 Bi 83 126 Be Mo 42 54arrow_forward
- Of the elements listed below, which one would be a poor conductor of heat and electricity? a) Mg b) Cs c) Se d) Sn e) Znarrow_forwardng GorillaGlass xB Wednesday-9/1 O Untitled presentation- ® HUNTER 7219768 Di X G There are only FIVE plax Pt Periodic Table- Plable x b web kamihq.com/web/viewer.html?state=%7B ids 3A%5B"IWjp_WcKxy3gFBs6la_nqTYuxT9-39na3%5 K A Classes Classkick O Remind E Edulastic icon O Nearpod icon Student Command O Google Keep A Desmos Scientiic G Soulhside High Sch. B Readin Pa. Edu O Pe Kami Uploads HUNTER 7219768 - Discovering the PT.pdf 257% they? 6. Which element has the longest name? Which element has the shortest name? 7. How many elements have a name ending in the suffix -ium &There are only FIVE places on the Periodic Table where pair of consecutive elements have the same SECOND Iletter in 16- their symbol. What are these five pairs? Which GROUP of elements has the suffix of -ine? D. What is the only element whose Average Atomic Mass is very close to its Atomic Number? 11. What element's name has another element's name founc within it? There are multiple situations-give at least one acerarrow_forwardStart Over (with same settings) Start Over View Results Read Explanation C Skip Question Question Number 4 of 20 - Chemistry How does the radioactive isotope C-14 differ from its stable counterpart C-12? It has a different number of protons and two less neutrons than C-12. It has the same number of protons and two more electrons than C-12. It has the same number of protons but two more neutrons than C-12. It has a different number of protons and two more neutrons than C-12.arrow_forward
- Atoms with the same number of protons but with different electrical charges _____.arrow_forward2.41 How does the periodic table help to make the study of chemistry more systematic?arrow_forwardYou perform a chemical reaction using the hypothetical elements A and B. These elements are represented by their molecular models shown below: The product of the reaction represented by molecular models is a Using the molecular models and the boxes, present a balanced chemical equation for the reaction of elements A and B. b Using the symbols A and B2 for the chemical reaction, write a balanced chemical equation. c What are some real-element possibilities for element B?arrow_forwardHundreds of years ago, alchemists tried to turn lead into gold. Is this possible? If not, why not? If yes, how would you do it?arrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
Recommended textbooks for you

 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY