
Concept explainers
(a)
Interpretation:
The ground state (or) the excited state electronic configuration of the given configuration has to be found.
Concept Introduction:
Ground state configurations:
Atoms may occupy different energy states. The energy states are distinct, they occur at specific values only. Therefore an atom can only move to new energy levels if it absorbs or emits light an amount of energy that exactly corresponds to the difference between two energy levels. The lowest possible energy that the atom can occupy is called the ground state. The arrangement of electrons in the lowest possible energy state is ground state electronic configuration.
Excited state configuration:
An excited state is an energy level of atom in which electron is at a higher energy level than its ground state.
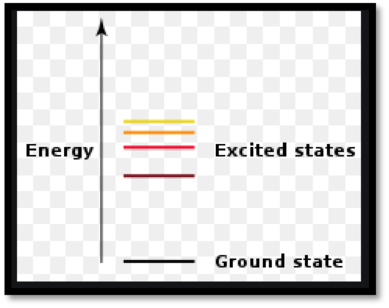
Figure 1
(b)
Interpretation:
The ground state (or) the excited state electronic configuration of the given configuration has to be found.
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The ground state (or) the excited state electronic configuration of the given configuration has to be found.
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
The ground state (or) the excited state electronic configuration of the given configuration has to be found.
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
General Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





