
Concept explainers
The following Lewis structures for (a) HCN, (b) C2H2, (c) SnO2, (d) BF3, (e) HOF, (f) HCOF, and (g) NF3 are incorrect. Explain what is wrong with each one and give a correct structure for the molecule. (Relative positions of atoms are shown correctly.)
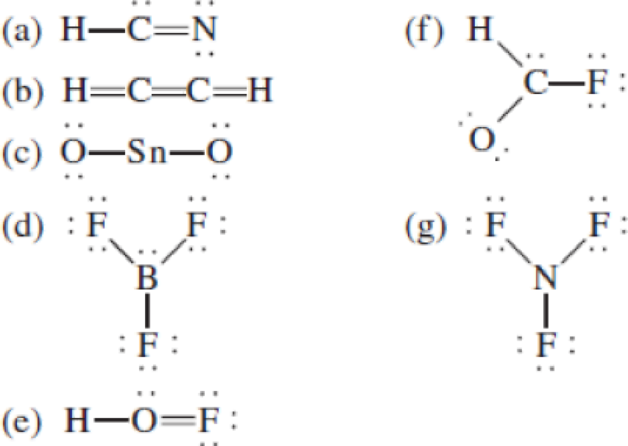
(a)
Interpretation: The Lewis structures of the molecules should be corrected with appropriate explanation.
Concept Introduction: Lewis structures are diagrams that represent the chemical bonding of covalently bonded molecules and coordination compounds.
It is also known as Lewis dot structures which represents the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule.
Dots represent the electron position around the atoms and lines or dot pairs represent covalent bonds between atoms.
The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell.
Answer to Problem 9.47QP
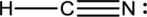
Explanation of Solution
To find: The correct Lewis structure for the given molecule.
- The given structure of the molecule is shown below.
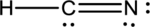
- In the given structure, the carbon contains lone pair of electrons and the bond between carbon and nitrogen is double bond. So the octets of these two atoms are not filled.
- The corrected Lewis structure of the above compound is drawn below.
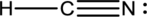
The total number of valence electrons is found to be 10, where 1 electron, 5 electrons and 4 electrons were contributed by H, C and N atoms respectively. Carbon is placed as the central atoms since its electronegativity is less than nitrogen.
The 6 electrons getting after reducing two electrons for each bond from the total valence electron are distributed on N atom to complete the octet. Sincethe octets of C atoms are not filled, a triple bond is made between C and N atoms.
(b)
Interpretation: The Lewis structures of the molecules should be corrected with appropriate explanation.
Concept Introduction: Lewis structures are diagrams that represent the chemical bonding of covalently bonded molecules and coordination compounds.
It is also known as Lewis dot structures which represents the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule.
Dots represent the electron position around the atoms and lines or dot pairs represent covalent bonds between atoms.
The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell.
Answer to Problem 9.47QP

Explanation of Solution
To find: The correct Lewis structure of the given molecule.
- The given structure of the molecule is below.

- In the given structure there is a double bond between hydrogen and carbon which violates the octet rule and also the bond between 2 carbon atoms is double.
- The corrected Lewis structure of the above compound is drawn below.

Each carbon atom bonded with one carbon and hydrogen atom. The total number of valence electrons found to be 10, where 1 electron, 5 electrons were contributed by each H and C atoms respectively.
Sincethere are noelectrons to distribute after reducing two electrons for each bond from the total valence electron, a triple bond is made between two C atomsto fill the octets.
(c)
Interpretation: The Lewis structures of the molecules should be corrected with appropriate explanation.
Concept Introduction: Lewis structures are diagrams that represent the chemical bonding of covalently bonded molecules and coordination compounds.
It is also known as Lewis dot structures which represents the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule.
Dots represent the electron position around the atoms and lines or dot pairs represent covalent bonds between atoms.
The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell.
Answer to Problem 9.47QP
Explanation of Solution
To find: The correct Lewis structure of the given molecule.
- The given structure of the molecule is below.
- In the given structure, tin atom does not fill the octet since the bond between tin and terminal atoms are single bond.
- The corrected Lewis structure of the above compound is drawn below.
The electronegativity of tin atom is less than oxygen, so it is taken as the central atom bonded with an oxygen atom at each side. Tin atom contributes 4 and each oxygen atom contributes 6 electrons making the total number of valence electrons 16.
To obtain the remaining electrons 12, two electrons for each bond is reduced from the total number of valence electrons, which then further distributed on the terminal oxygen atoms to fill the octets.
Since the central tin atom does not complete octet, a double bond is formed between each terminal oxygen atom
(d)
Interpretation: The Lewis structures of the molecules should be corrected with appropriate explanation.
Concept Introduction: Lewis structures are diagrams that represent the chemical bonding of covalently bonded molecules and coordination compounds.
It is also known as Lewis dot structures which represents the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule.
Dots represent the electron position around the atoms and lines or dot pairs represent covalent bonds between atoms.
The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell.
Answer to Problem 9.47QP
Explanation of Solution
To find: The correct Lewis structure of the given molecule.
- The given structure of the molecule is below.
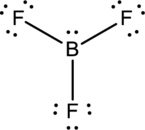
- In this structure, there is a lone pair of electron on boron atom whereas in actual structure that lone pair is no needed to fill the octet.
- The corrected Lewis structure of the above compound is drawn below.
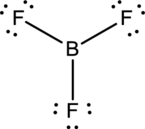
Boron atom has less electronegativity comparing to fluorine. So it is taken as the central atom with 3 terminal fluorine atoms.The boron has 4 and each fluorine atom have 7 valence electrons. Since there are 3 fluorine atoms the total valence electron of the molecule becomes 24.
The 18 electrons after reducing two electrons for each bond from the total valence electron are distributed onfluorine atom to fill the octets. So each fluorine atom gets 3 lone pairs.
(e)
Interpretation: The Lewis structures of the molecules should be corrected with appropriate explanation.
Concept Introduction: Lewis structures are diagrams that represent the chemical bonding of covalently bonded molecules and coordination compounds.
It is also known as Lewis dot structures which represents the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule.
Dots represent the electron position around the atoms and lines or dot pairs represent covalent bonds between atoms.
The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell.
Answer to Problem 9.47QP
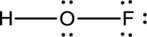
Explanation of Solution
To find: The correct Lewis structure of the given molecule
- The given structure of the molecule is below.
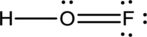
- In the given structure, there is a double bond between oxygen and fluorine which is not needed. Distributing a lone pair on oxygen is enough to fill its octet.
- The corrected Lewis structure of the above compound is drawn below.
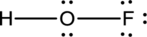
The electronegativity of oxygen atom is less than fluorine and the molecule is with hydrogen and fluorine atoms at the terminal position of oxygen.
Hydrogen, oxygen and fluorine contribute 1, 6 and 7 electrons respectively making the total number of valence electrons 14.
The 10 electrons after reducing for each bond from the total valence electron are distributed on terminal atoms, then to central oxygen atom to fill the octets.
(f)
Interpretation: The Lewis structures of the molecules should be corrected with appropriate explanation.
Concept Introduction: Lewis structures are diagrams that represent the chemical bonding of covalently bonded molecules and coordination compounds.
It is also known as Lewis dot structures which represents the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule.
Dots represent the electron position around the atoms and lines or dot pairs represent covalent bonds between atoms.
The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell.
Answer to Problem 9.47QP
Explanation of Solution
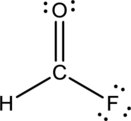
- The given structure of the molecule is below.
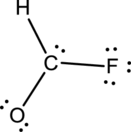
- Here the bond between oxygen and carbon is not given appropriately to fill the octet.
- The corrected Lewis structure of the above compound is drawn below.
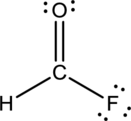
Comparing to fluorine and oxygen, carbon has the least electronegativity, so it is taken as the central atom with hydrogen, fluorine and oxygen at its terminal positions.
Hydrogen, oxygen, carbon and fluorine contribute 1, 6, 4 and 7 electrons respectively making the total number of valence electrons 18.
To fill the octets of the atoms, the 12 electrons after reducing two electrons for each bond from the total valence electron are distributed on terminal atoms.
Since the central carbon atom does not have sufficient electrons to fill the octet, a double bond is made between carbon and terminal oxygen atoms.
(g)
Interpretation: The Lewis structures of the molecules should be corrected with appropriate explanation.
Concept Introduction: Lewis structures are diagrams that represent the chemical bonding of covalently bonded molecules and coordination compounds.
It is also known as Lewis dot structures which represents the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule.
Dots represent the electron position around the atoms and lines or dot pairs represent covalent bonds between atoms.
The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell.
Answer to Problem 9.47QP
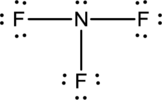
Explanation of Solution
- The given structure of the molecule is below.
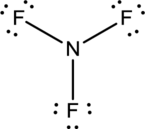
- In this structure a lone pair of electrons is missing which is sufficient to fill the octet of nitrogen.
- The corrected Lewis structure of the above compound is drawn below.
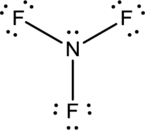
Nitrogen atom has less electronegativity comparing to fluorine. So it is taken as the central atom with 3 fluorine atoms at the terminal positions of it
The nitrogen has 5 and each fluorine atom have 7 valence electrons. Since there are 3 fluorine atoms the total number of valence electrons becomes 26.
The 20 electrons after reducing two electrons for each bond from the total valence electron are distributed on fluorine atom to fill the octets. The remaining 2 electrons are distributed to the central atom so that nitrogen fills the octet.
Want to see more full solutions like this?
Chapter 9 Solutions
Chemistry
- The study of carbon-containing compounds and their properties is called organic chemistry. Besides carbon atoms, organic compounds also can contain hydrogen, oxygen, and nitrogen atoms (as well as other types of atoms). A common trait of simple organic compounds is to have Lewis structures where all atoms have a formal charge of zero. Consider the following incomplete Lewis structure for an organic compound called histidine (an amino acid), which is one of the building blocks of proteins found in our bodies: Draw a complete Lewis structure for histidine in which all atoms have a formal charge of zero.arrow_forwardGiven the bonds C N, C H, C Br, and S O, (a) which atom in each is the more electronegative? (b) which of these bonds is the most polar?arrow_forwardUse Lewis structures to determine which two of the follow-ing are unstable: (a) SF₂; (b) SF₃; (c) SF₄; (d) SF₅; (e) SFarrow_forward
- Draw a Lewis structure for (a) PF₃; (b) H₂CO₃(both H atoms are attached to O atoms); (c) CS₂.arrow_forward8) Draw complete Lewis structures and indicate, using VSEPR, which of the following species (there may be more than one) is/are likely to have the see-saw molecular shape: (a) XeF4, (b) BrF4*, (c) SiF4, (d) TeCl4, (e) HCIO4 (f) Xe04?arrow_forwardWrite the Lewis symbol for atoms of each of the following elements:(a) Al, (b) Br, (c) Ar, (d) Sr.arrow_forward
- 3) Answer the following questions using principles of chemical bonding and molecular structure: (a) Consider the molecules CF4 and SF4. (i) Draw the complete Lewis electron-dot structure for each molecule. (ii) In terms of molecular geometry, account for the fact that the CF4 molecule is nonpolar, whereas the SF4 molecule is polar.arrow_forwardNASA’s Cassini-Huygens mission detected a large cloud of toxic hydrogen cyanide (HCN) on Titan, one of Saturn’s moons. Titan also contains ethane (H3CCH3), acetylene (HCCH), and ammonia (NH3). What are the Lewis structures of these molecules?arrow_forwardWrite Lewis structures for the following molecules and ions: (a) NCl3, (b) OCS, (c) H2O2arrow_forward
- carbon tetrachloride was formerly used in fire extinguishers for electrical fires.it is no longer used for this purpose because of the formation of the toxic gas phosgene,CL2CO.wRITE THE LEWIS STRUCTURES for the methane and ethane molecules.arrow_forwardConsider peroxynitrite (chemical formula: ONOO−), a structural isomer of the nitrate anion. It is generated in the cell when nitric oxide (NO) reacts with the superoxide radical anion (O2-.). Peroxynitrite is a potent oxidant and nitrating agent and can lead to DNA and protein damage (this is the complete question) Draw the correct Lewis structure for peroxynitrite and indicate approximate bond angles. Tell how many molecular degrees of freedom of motion are present in peroxynitrite and what motions they correspond to.arrow_forwardCarbon tetrachloride was formerly used in fire extinguishers for electrical fires. It is no longer used for this purpose because of the formation of the toxic gas phosgene, Cl2CO. Write the Lewis structures for carbon tetrachloride and phosgene.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning




