
Concept explainers
(a)
To determine: The structure for the given compound.
Interpretation: The structure for the given compound is to be drawn.
Concept introduction:
(a)
Answer to Problem 7.40SP
The structure of
Explanation of Solution
The given compound is
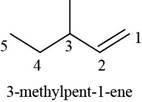
Figure 1
(b)
To determine: The structure for the given compound.
Interpretation: The structure for the given compound is to be drawn.
Concept introduction: Alkenes and alkynes are unsaturated hydrocarbons and are named by identifying the longest chain of carbon atoms that contain double or triple bond, their names end by adding suffix –ene and –yne to the parent hydrocarbon.
(b)
Answer to Problem 7.40SP
The structure of
Explanation of Solution
The given compound is
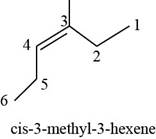
Figure 2
(c)
To determine: The structure for the given compound.
Interpretation: The structure for the given compound is to be drawn.
Concept introduction: Alkenes and alkynes are unsaturated hydrocarbons and are named by identifying the longest chain of carbon atoms that contain double or triple bond, their names end by adding suffix –ene and –yne to the parent hydrocarbon.
(c)
Answer to Problem 7.40SP
The structure of
Explanation of Solution
The given compound is
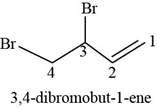
Figure 3
(d)
To determine: The structure for the given compound.
Interpretation: The structure for the given compound is to be drawn.
Concept introduction: Alkenes and alkynes are unsaturated hydrocarbons and are named by identifying the longest chain of carbon atoms that contain double or triple bond, their names end by adding suffix –ene and –yne to the parent hydrocarbon.
(d)
Answer to Problem 7.40SP
The structure of
Explanation of Solution
The given compound is
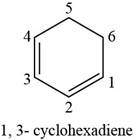
Figure 4
(e)
To determine: The structure for the given compound.
Interpretation: The structure for the given compound is to be drawn.
Concept introduction: Alkenes and alkynes are unsaturated hydrocarbons and are named by identifying the longest chain of carbon atoms that contain double or triple bond, their names end by adding suffix –ene and –yne to the parent hydrocarbon.
(e)
Answer to Problem 7.40SP
The structure of
Explanation of Solution
The given compound is
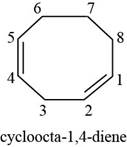
Figure 5
(f)
To determine: The structure for the given compound.
Interpretation: The structure for the given compound is to be drawn.
Concept introduction: Alkenes and alkynes are unsaturated hydrocarbons and are named by identifying the longest chain of carbon atoms that contain double or triple bond, their names end by adding suffix –ene and –yne to the parent hydrocarbon.
(f)
Answer to Problem 7.40SP
The structure of
Explanation of Solution
The given compound is
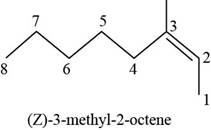
Figure 6
(g)
To determine: The structure for the given compound.
Interpretation: The structure for the given compound is to be drawn.
Concept introduction:
(g)
Answer to Problem 7.40SP
The structure of vinylcyclopropane is shown in Figure 7.
Explanation of Solution
The given compound is vinylcyclopropane. The root name cyclopropane signifies three carbon atoms in a ring. Vinyl group is the
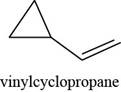
Figure 7
(h)
To determine: The structure for the given compound.
Interpretation: The structure for the given compound is to be drawn.
Concept introduction: Alkenes and alkynes are unsaturated hydrocarbons and are named by identifying the longest chain of carbon atoms that contain double or triple bond, their names end by adding suffix –ene and –yne to the parent hydrocarbon.
(h)
Answer to Problem 7.40SP
The structure of
Explanation of Solution
The given compound is
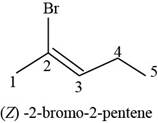
Figure 8
(i)
To determine: The structure for the given compound.
Interpretation: The structure for the given compound is to be drawn.
Concept introduction: Alkenes and alkynes are unsaturated hydrocarbons and are named by identifying the longest chain of carbon atoms that contain double or triple bond, their names end by adding suffix –ene and –yne to the parent hydrocarbon.
(i)
Answer to Problem 7.40SP
The structure of
Explanation of Solution
The given compound is
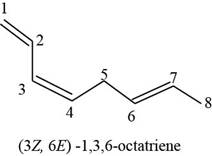
Figure 9
Want to see more full solutions like this?
Chapter 7 Solutions
Organic Chemistry (9th Edition)
- Reagents a. C6H5CHO b. NaOH, ethanol h. BrCH2CH=CH2 i. Na* OEt, ethanol j. Br2, H* k. K* t-BuO c. Pyrrolidine, cat. H* d. H2C=CHCN e. H3O* f. I. CH2(CO2ET)2 -CH2CH2CN LDA m. heat g. ELOC(=0)CO2ET Select reagents from the table to synthesize this compound from cyclopentanone. Enter the letters of the chosen reagents, in the order that you wish to use them, without spaces or punctuation (i.e. geda).arrow_forwardDraw the skeletal structures for the compounds in a. (Z)-1,3,5-tribromo-2-pentene b. (Z)-3-methyl-2-heptenec. (E)-1,2-dibromo-3-isopropyl-2-hexene d. vinyl bromidee. 1,2-dimethylcyclopentene f. diallylaminearrow_forward1. Give the structure corresponding to each name. a. 4-ethyl-3-heptanol b. 3-chloro-1,2-propanediol c. diisobutyl ether d. 1,2-epoxy-1,3,3-trimethylcyclohexane e. 1-ethoxy-3-ethylheptanearrow_forward
- 3. Draw the structure of the following alkenes. Some of these compounds can show isomerism, and some cannot. Indicate which among these can show cis and trans isomers. a. hex-3-ene b. buta-1,3-diene c. 2,3-dimethylpent-2-ene 4. Draw the structure of the following alkenes. Some of these compounds can show isomerism, and some cannot. Indicate which among these can show cis and trans isomers. a. 3-ethylhexa-2,4-diene b. pent-1,3-diene c. 3,7-dichloroocta-2,5-dienearrow_forwardShow how to convert propene to each of these compounds, using any inorganic reagents as necessary. a. Propane b.1,2-Propanediol c. 1-Propanol d. 2-Propanol e. Propanal f. Propanone g. Propanoic acid h. l-Bromo-2-propanol i. 3-Chloropropene j. 1,2,3-Trichloropropane k. 1-Chloropropane l. 2-Chloropropane m. 2-Propen-1-ol n. Propenalarrow_forwarda. 4-bromopentyne b. 1-bromo-2-pentyne c. 1-bromo-3-pentyne d. 4-bromo-2-pentynearrow_forward
- These compounds are unreactive to NaNH2 EXCEPT a. 1-butene. b. 2-butyne. c. 2-butene. d. 1-butyne.arrow_forward12. Write structural formulas for the following alkene names. a. hept-2-ene b. hex-2-ene 6,7-dimethyloct-1-ene d. 2,4-dimethyloct-4-ene с. 3-ethylnon-2-ene е.arrow_forward7. Draw the structure for each compound. A. acetylene B 3-methyl-1-hexyne C. 4-methyl-2-hexyne D. 3-octynearrow_forward
- Please use either full structural diagrams or the combination method shown in the lesson. Skeletal/line diagrams will not be accepted. a. 5-butyl-4-ethyl-6- methylhept-3 - ene b. 3-propyl-2-methylcyclohept-1 - ene c. 2, 5, 5, 6-tetramethylhept-3-yne d. 1-ethyl-3- propylbenzenearrow_forward4 Which involve large number of organic reaction donot which involve large number of small molecules called 8. involve and free radicals O A. Addition reaction. B. Substitution reaction. OC. Elimination reaction. O D. Pericyclic reaction O E. Rearrangement reaction. 9. monomers to from molecule high molecule weigh. A. Substitution reaction. B. Elimination reaction. C. Polymerization reaction. D. Pericyclic reaction.arrow_forwarddocs.google.com 3. Substation reaction is define as. A. Replaced by another atom or group of atoms. B. Added to a molecule with multiple bond. C. Loss of two atoms and groups. is opposite of. 4. OA. Addition reaction. B. Substation reaction C. Elimination reaction. 5. Method is preparing compounds with double and triple bonds is. A. Elimination reaction. B. Substation reaction C. Addition reaction.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





