
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10, Problem 10.31SP
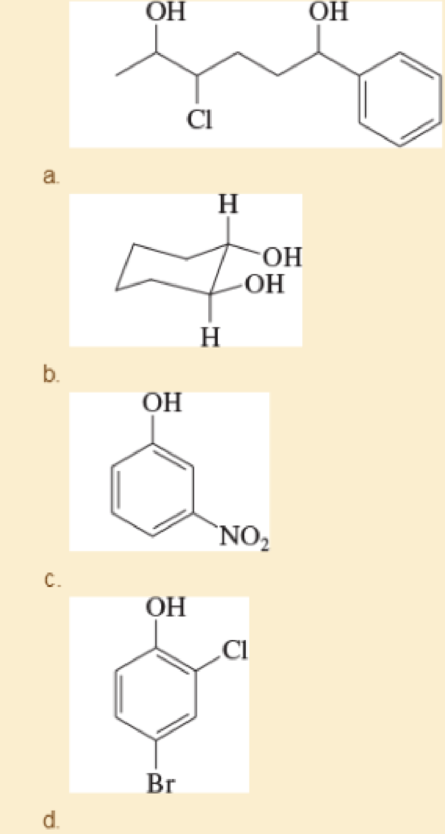
Give systematic (IUPAC) names for the following
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Write the chemical equation showing reactants, products and catalysts needed (if any) for thefollowing reactions. Write the IUPAC name of the product right beside the structure.
a) Reaction of two phenol
b) Reaction of 2-bromophenol with sodium hydroxide
c) Reaction of potassium phenoxide with 2-chloropentane
d) Reaction of isopropyl propyl ether with HBr
e) Reaction of 3-methylpentan-3-ol with sulfuric acid
f) Dehydration of 1,5-hepta-diolg) Oxidation of 1-decanolh) Hydration od hepta-2,3-diene
When trans-2-chloro-1-cyclohexanol is treated with a base, cyclohexene oxide is the product. However, when cis-2-chloro-1-cyclohexanol is treated with a base, the product is cyclohexanone i. Write the equation for the reaction between trans-2-chloro-1-cyclohexanol and the base to yield the cyclohexene oxide.
ii. Why doesn’t the cis isomer yield the oxide?.
iii. Write the mechanism for each of the two reactions.
for the following compounds, give their IUPAC and common names:
Chapter 10 Solutions
Organic Chemistry (9th Edition)
Ch. 10.3A - Prob. 10.1PCh. 10.3B - Give both the IUPAC name and the common name for...Ch. 10.3B - Prob. 10.3PCh. 10.3C - Give a systematic (IUPAC) name for each diol. a....Ch. 10.4B - Predict which member of each pair will be more...Ch. 10.4B - Dimethylamine (CH3)2NH, has a molecular weight of...Ch. 10.6A - Prob. 10.7PCh. 10.6A - Prob. 10.8PCh. 10.6C - A nitro group (NO2) effectively stabilizes a...Ch. 10.6C - Prob. 10.10P
Ch. 10.8B - Prob. 10.11PCh. 10.8B - Prob. 10.12PCh. 10.9A - Prob. 10.13PCh. 10.9B - Prob. 10.14PCh. 10.9C - Show how you would synthesize each tertiary...Ch. 10.9D - Prob. 10.16PCh. 10.9D - Show how you would add Grignard reagents to acid...Ch. 10.9D - A formate ester, such as ethyl formate, reacts...Ch. 10.9E - Prob. 10.19PCh. 10.9E - In Section9-7B, we saw how acetylide ions add to...Ch. 10.9F - Prob. 10.21PCh. 10.10A - Prob. 10.22PCh. 10.10B - Prob. 10.23PCh. 10.11B - Predict the products you would expect from the...Ch. 10.11B - Prob. 10.25PCh. 10.11B - Prob. 10.26PCh. 10.12 - Prob. 10.27PCh. 10.12 - Prob. 10.28PCh. 10.12 - Authentic skunk spray has become valuable for use...Ch. 10 - Give a systematic (IUPAC) name for each alcohol....Ch. 10 - Give systematic (IUPAC) names for the following...Ch. 10 - Draw the structures of the following compounds...Ch. 10 - Predict which member of each pair has the higher...Ch. 10 - Predict which member of each pair is more acidic,...Ch. 10 - Predict which member of each group is most soluble...Ch. 10 - Draw the organic products you would expect to...Ch. 10 - Prob. 10.37SPCh. 10 - Show how you would synthesize the following...Ch. 10 - Show how you would use Grignard syntheses to...Ch. 10 - Show how you would accomplish the following...Ch. 10 - Show how you would synthesize the following: a....Ch. 10 - Complete the following acid-base reactions. In...Ch. 10 - Prob. 10.43SPCh. 10 - Prob. 10.44SPCh. 10 - Geminal diols, or 1,1-diols, are usually unstable,...Ch. 10 - Vinyl alcohols are generally unstable, quickly...Ch. 10 - Compound A (C7H11Br) is treated with magnesium in...Ch. 10 - Prob. 10.48SPCh. 10 - Prob. 10.49SPCh. 10 - Prob. 10.50SPCh. 10 - Prob. 10.51SPCh. 10 - Prob. 10.52SPCh. 10 - Prob. 10.53SPCh. 10 - Prob. 10.54SPCh. 10 - Prob. 10.55SPCh. 10 - Prob. 10.56SPCh. 10 - Show how this 1 alcohol can be made from the...Ch. 10 - Prob. 10.58SPCh. 10 - Prob. 10.59SPCh. 10 - Prob. 10.60SP
Additional Science Textbook Solutions
Find more solutions based on key concepts
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Give one example from main group chemistry that illustrates each of the following descriptions: (a) Covalent ne...
General Chemistry: Atoms First
Practice Problem ATTEMPT
Write the rate expressions for each of the following reactions:
(a)
(b)
(c)
Chemistry
2. Why shouldn’t you work in a laboratory by yourself?
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (14th Edition)
Describe the orbitals used in bonding and the bond angles in the following compounds: a. CH3O b. CO2 c. H2CO d....
Organic Chemistry (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Give IUPAC names for the following substances:arrow_forwardGive the IUPAC name of the alkyne that produces only propanoic acid (CH3CH2CO2H) when treated with ozone followed by hydrolysis.arrow_forwardComplete the following syntheses – they may be two- or three-step processes. Include any necessary catalysts or reaction conditions. a) Prepare propanone from 1-propanolarrow_forward
- When trans-2-chloro-1-cyclohexanol is treated with a base, cyclohexene oxide is the product. However, when cis-2-chloro-1-cyclohexanol is treated with a base, the product is cyclohexanoneWrite the equation for the reaction between trans-2-chloro-1-cyclohexanol and the base to yield the cyclohexene oxide Why doesn’t the cis isomer yield the oxide?Write the mechanism for each of the two reactions. .arrow_forwardCompare the families of alcohols, phenols, ethers, thiols and alkyl halides in terms of polarity, acidity, reactivity, hydrogen bond formation, relative boiling points.arrow_forwardALCOHOLS, PHENOLS AND ETHERSarrow_forward
- Name: Organic Chemistry Nomenclature Project Esters CH3 H,C- propyl butanoate phenyl hexanoate dichloromethyl-2-ethoxypropanoate Br CH3 H,C CH3 methyl propanotate CH3 H3C CH3 2- nitrohexyl ethanoatearrow_forwardAldehydes and ketones are soluble in organic solvents True False The IUPAC name of the compound CH3CH2COCH3 is ******** The reaction between strong base and C6H6Cl is called The reaction between phenol and nitric acid product O-nitro phenol p-nitro phenol picric acid 2,4-nitrophenol إجابتك إجابتكarrow_forwardGive the structures and IUPAC names for all tertiary alcohols of molecular formula C6H10O.arrow_forward
- Isoamyl acetate is the common name of the substance most responsible for the characteristic odor of bananas. Write a structural formula for isoamyl acetate, given the information that it is an ester in which the carbonyl group bears a methyl substituent and there is a 3-methylbutyl group attached to one of the oxygens.arrow_forwardGive IUPAC name for the following compound:arrow_forwardAlcohols undergo dehydration reactions in the presence of an acid catalyst. Which of the following compounds yields only a single alkene product upon dehydration?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY