
(a)
Interpretation: The structure of the major product formed from acid-catalyzed hydration of
Concept introduction:Acid-catalyzed hydration is the electrophilic addition of water. The reactive species that act as a catalyst is
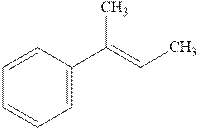
(b)
Interpretation: The structure of the major product formed fromacid-catalyzed hydration alkene indicated should be written.
Concept introduction:Acid-catalyzed hydration is the electrophilic addition of water. The reactive species that act as a catalyst is
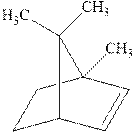
(c)
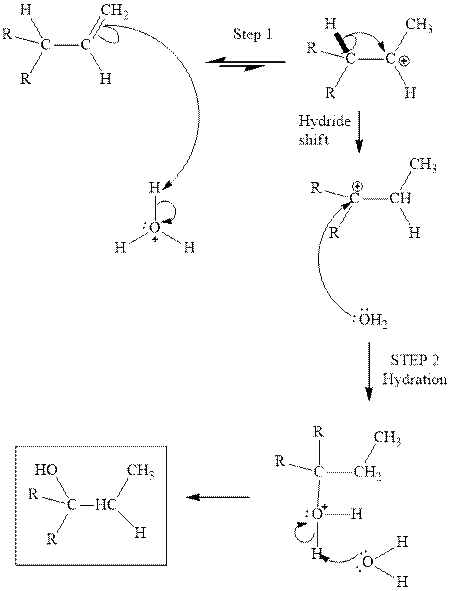
Interpretation: The structure of the major product formed from acid-catalyzed hydration alkene indicated should be written.
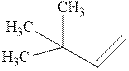
Concept introduction:Acid-catalyzed hydration is the electrophilic addition of water. The reactive species that act as a catalyst is
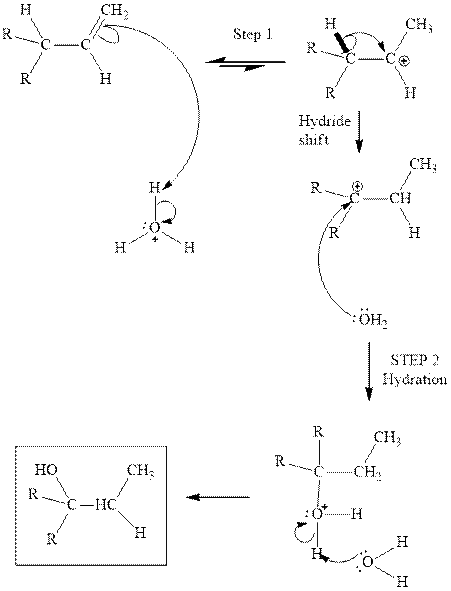
(d)
Interpretation: The structure of the major product formed from acid-catalyzed hydration of alkene indicated should be written.
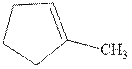
Concept introduction:Acid-catalyzed hydration is the electrophilic addition of water. The reactive species that act as a catalyst is
in the first stage. In the second stage water itself acts as a nucleophile and another water abstracts a proton to give final hydration product as illustrated below.
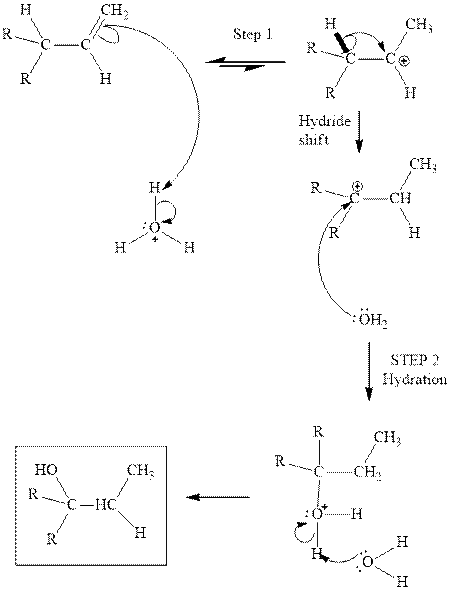
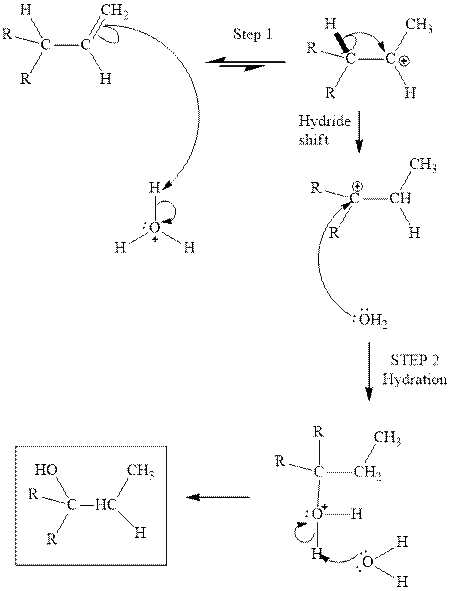
(d)
Interpretation: The structure of the major product formed from acid-catalyzed hydration alkene indicated should be written.
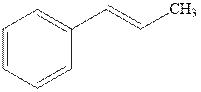
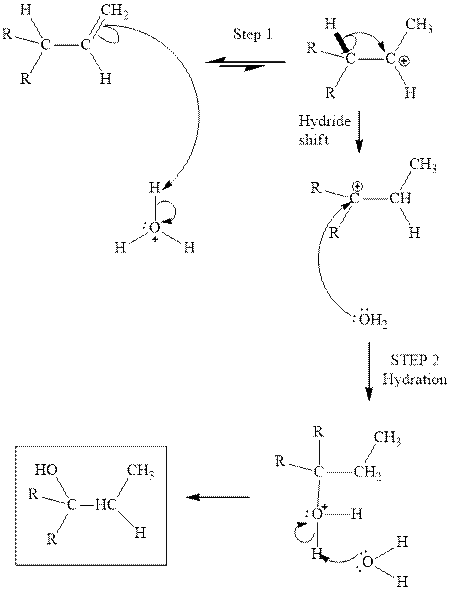
Concept introduction:Acid-catalyzed hydration is the electrophilic addition of water. The reactive species that act as a catalyst is
in the first stage. In the second stage water itself acts as a nucleophile and another water abstracts a proton to give final hydration product as illustrated below.
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
EBK EXPERIMENTAL ORGANIC CHEMISTRY: A M
- Discuss and explain in detail what phenols are, its properties, the different tests used to identify them and compare phenols to alcohols, its properties and lucas test conducted to compare the reactivity of primary, secondary and tertiary alcohols.arrow_forwardDraw structural formulas for the alkene that gives each alcohol upon hydroboration-oxidation.arrow_forwardDraw structural formulas for the alkenes formed by acid-catalyzed dehydration of each alcohol. Where isomeric alkenes are possible, predict which is the major product. Q.) 1-Methylcyclopentanolarrow_forward
- 1) The carbon-oxygen double bond present in aldehydes and ketones is very polar. What does this mean and how does it arise? 2) The carbon-oxygen double bond is readily attacked by nucleophiles like cyanide ions or ammonia. (i) What do you understand by the term nucleophile? (ii) Which part of the carbon-oxygen double bond is attractive to nucleophiles? 3) Why is there a difference between aldehydes and ketones in their response to oxidizing agents such as potassium dichromate(VI) solution acidified with dilute sulfuric acid?arrow_forwardWhat two alkenes give rise to attached alcohol as the major product of acid-catalyzed hydration?arrow_forwardWrite the products formed as a result of the reactions given below.arrow_forward
- What two alkenes give rise to each alcohol as the major product of acid-catalyzed hydration?arrow_forward-Draw the structural formulae of compounds Q, S and T. -The structural formula and IUPAC name for alcohol P. -The conditions and reagents needed for the reaction(i) to occur.arrow_forwardAldehydes and ketones react with one molecule of an alcohol to form compounds called hemiacetals, in which there is one hydroxyl group and one ether-like group. Reaction of a hemiacetal with a second molecule of alcohol gives an acetal and a molecule of water. We study this reaction in Chapter 16. Draw structural formulas for the hemiacetal and acetal formed from these reagents. The stoichiometry of each reaction is given in the problem.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
