
(a)
Interpretation:
Similarities and differences in the structures of aspirin, ibuprofen and acetaminophen have to be identified.
Concept Introduction:
Ibuprofen is a nonsteroidal anti-inflammatory drug (NSAID) used for the treatment of fever and inflammation. Side effects commonly seen for ibuprofen are bloating, headache, nausea etc. The structure of ibuprofen is,
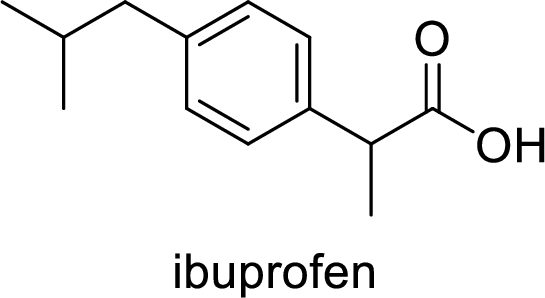
Acetaminophen used for the treatment of fever and pain. Side effects commonly seen for acetaminophen are jaundice, nausea, dark urine. The structure of acetaminophen is,
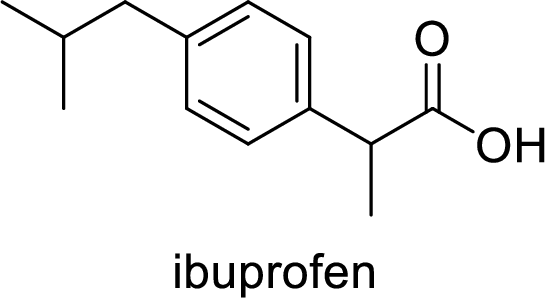
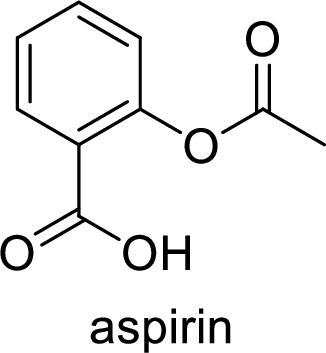
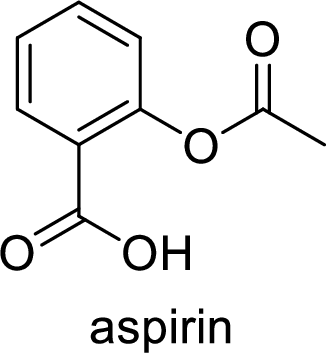
Aspirin used for the treatment of inflammation, fever and pain. Side effects commonly seen for aspirin are stomach ache, nausea and rash. The structure of aspirin is,
Alcohol: It is an organic compound where it contains at least one
(b)
Interpretation:
To find whether there is any similarities or differences in the symptoms that aspirin, ibuprofen and acetaminophen is promoted to treat have to be identified.
Concept Introduction:
Ibuprofen is a nonsteroidal anti-inflammatory drug (NSAID) used for the treatment of fever and inflammation. Side effects commonly seen for ibuprofen are bloating, headache, nausea etc. The structure of ibuprofen is,
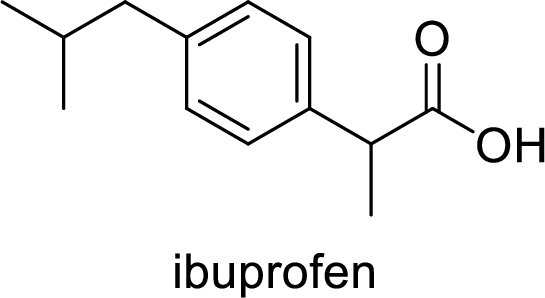
Acetaminophen used for the treatment of fever and pain. Side effects commonly seen for acetaminophen are jaundice, nausea, dark urine. The structure of acetaminophen is,
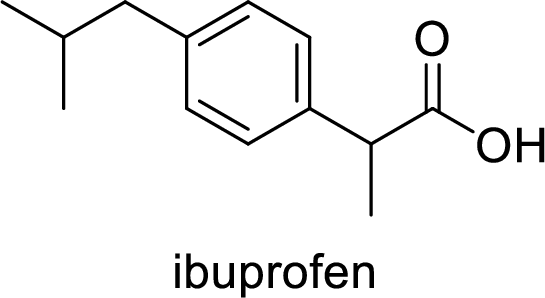
Aspirin used for the treatment of inflammation, fever and pain. Side effects commonly seen for aspirin are stomach ache, nausea and rash. The structure of aspirin is,
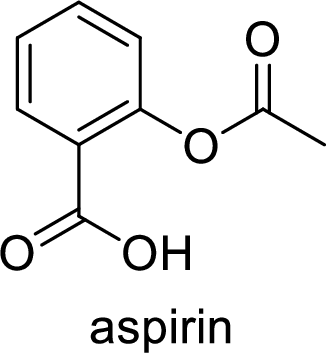
(b)
Interpretation:
To find whether there is any similarities or differences in the side effects of aspirin, ibuprofen and acetaminophen have to be identified.
Concept Introduction:
Ibuprofen is a nonsteroidal anti-inflammatory drug (NSAID) used for the treatment of fever and inflammation. Side effects commonly seen for ibuprofen are bloating, headache, nausea etc. The structure of ibuprofen is,
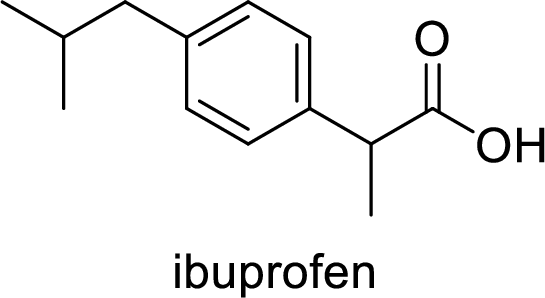
Acetaminophen used for the treatment of fever and pain. Side effects commonly seen for acetaminophen are jaundice, nausea, dark urine. The structure of acetaminophen is,
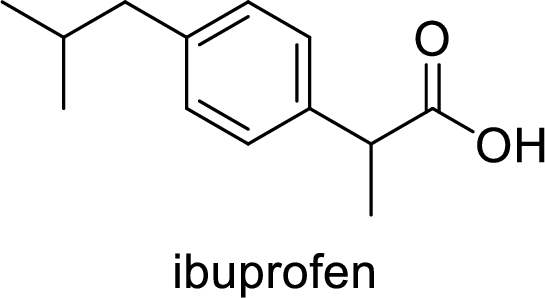
Aspirin used for the treatment of inflammation, fever and pain. Side effects commonly seen for aspirin are stomach ache, nausea and rash. The structure of aspirin is,
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Chemistry In Context
- Directions: Read each question carefully. Choose the letter of the correct answer. 1. Who were the first people to believe that soap-like material can treat skin diseases? a. Babylons 2. Why did soap-making become widespread in the 18th century? a. It was known to treat skin diseases. b. It was used for laundering and washing with much less effort. c. It was known as a luxurious cleaning agent among wealthy people. d. It was used to advertise campaigns on personal hygiene and health. 3. Which of the following is NOT an ingredient in soap-making? b. Egyptian c. Greeks d. Romans c. natural raw material d. vegetable oils a. animal fats b. hydrocarbon from petroleum 4. Which of the following forms scum or film when used in hard water? a. detergent 5. What alkali can be added to make hard soap? a. Caustic potash 6. What is the use of the hydrophobic end of the chemical composition of soap and detergent? b. soap c. both a and b d. none of the choices b. soap c. Potassium hydroxide d. Soda…arrow_forwardQuestion • Choose a pharmaceutical drug and answer the following questions about the drug: o What is the IUPAC chemical name? o Who discovered it or what company discovered it and when (if available)? o What does the compound look like? Provide a picture of the compound. o What are the functional groups in the compound? o What are the medical uses for the drug? o What is the mechanism of action of the drug (how does it work to affect the disease or malady)? o How is it usually administered (ex. IV, pill, etc.)? o What are the potential adverse effects of the drug? Expert Answer Step 1 Note - Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for you. To get remaining sub-part solved please repost the complete question and mention the sub-parts to be solved. A pharmaceutical drug used for treatment of fever and for mild to moderate pain is Paracetamol. Its molecular formulaarrow_forward7. Statement 1: All food additives are carcinogenic. Statement 2: Food additives must be avoided as much as possible. A. True, False B. True, True C. False, False D. False, True 8. How do thickeners improve a food product? A. They prevent spoilage due to oxygenation B. They increase the viscosity of a product C. They preserve the flavor of a product D. They keep food from drying out. _9. It makes the food thicker by keeping oil and water mixed together. A. Emulsifiers C. Humectants B. Flavor Enhancer D. Antioxidantsarrow_forward
- 1. Choose a molecule from a drug product or drug. 2. Indicate the common (industrial) name and its name according to IUPAC. 3. Draw the molecule. 4. Identify the functional groups of the molecule. 5. Discuss the role of these functional groups in the functioning of the molecule. 6. Explain the role and/or functioning of the molecule. 7. Analyze the risks and benefits of using the product. 8. Talk about the origin of the molecule as well as the stages of its synthesis (exemple: the number of steps, the types of reactions involved, other peculiarities).arrow_forward1. The Ability of a substance to exist in different crystalline form a. Lattice b. Polymorphism c. Crystallization d. Amphoterism 2. A drug can exert its pharmacological effect only if it is a. Protein bound b. Protein unbound c. Free drug d. Both B & C e. Both A & C 3. In order for the drug to be ready and available for absorption, it must be release first from its dosage form with the exception of: a. Capsule b. Tablet c. Solution d. Suspension 4. All of the following are true, except a. Solubility increase with decrease particle size b. Solubility increase with increase surface area c. Solubility increase with increase particle size d. Solubility decrease with decrease surface area 5. The rate in which the drug appears in the bloodstream is also known as a. Half-life b. Potency c. Bioavailability d. Area under the curvearrow_forwardamine 63. This picture is an example of beverages." alcoholic fruity citrusarrow_forward
- 1. Differentiate volatile oil from fixed oil (table)2. Which among the crude volatile oil drugs exhibit a hallucinogenic (amphetamine-like) properly and is commonly used by prison inmates.arrow_forwardWhich of the two is more effective in disinfecting our hands especially this time of pandemic? a. 70 % ethyl alcohol b. 30% ethyl alcohol c. 40% Isopropyl alcohol. d. 75% isopropyl alcoholarrow_forwardHi! I need help with describing the structure of this drug Flexeril also know as Cyclobenzaprine. How would you give your audience information on this structure of this drug in a powerpoint presentation? Thank you.arrow_forward
- Based on a grade 11 chemistry student, answer the following question based on the provided tables: Part G: Final Identification of the Poison By this time, Tylenol has been pulled off the shelves at all of the stores in the area and a nationwide recall has been announced. Several suspicious capsules have been found and tested. The contents of the capsules contain a compound with a nitrogen content of greater than 15% and a carbon content of less than 50%. What is the poison in the capsule? Provide calculations to support your answer.arrow_forward1. What are the measures that can help prevent proliferation of counterfeit drugs in the Philippines? 2. What is the role of a pharmacist (practicing in other areas other than the community) in resolving this problem?arrow_forwardWhat is the importance of knowing the chemistry of drugs?arrow_forward

 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry In FocusChemistryISBN:9781305084476Author:Tro, Nivaldo J., Neu, Don.Publisher:Cengage Learning
Chemistry In FocusChemistryISBN:9781305084476Author:Tro, Nivaldo J., Neu, Don.Publisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





