
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13, Problem 13.53SP
Each of these four structures has molecular formula C4cH8O2. Match the structure with its characteristic proton NMR signals. (Not all of the signals are listed in each case.)
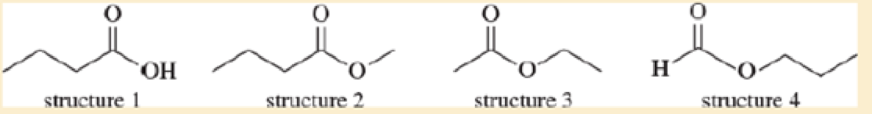
- a. sharp 1H singlet at δ 8.0 and 2H triplet at δ 4.0
- b. sharp 3H singlet at δ 2.0 and 2H quartet at δ 4.1
- c. sharp 3H singlet at δ 3.7 and 2H quartet at δ 2.3
- d. broad 1H singlet at δ 11.5 and 2H triplet at δ 2.3
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Construct a structure of the given 1H NMR data signals.A.
C4H11N
0.94 δ (3H, triplet)
1.50 δ (2H, sextet)
1.69 δ (1H, singlet)
2.40 δ (3H, singlet)
2.55 δ (2H, triplet)
B.
C9H11Br
2.15 δ (2H, quintet, J = 7 Hz)
2.75 δ (2H, triplet, J = 7 Hz)
3.38 δ (2H, triplet, J = 7 Hz)
7.22 δ (5H, multiplet)
1Compound 1 has molecular formula C7H16. It shows three signals in the 1H-NMR spectrum, one at 0.85 ppm, one at 1.02 ppm, and one at 1.62 ppm. The relative integrals of these three signals are 6, 1, and 1, respectively.
Compound 2 has molecular formula C7H14. It shows three signals in the 1H-NMR spectrum, one at 0.98 ppm, one at 1.36 ppm, and one at 1.55 ppm. The relative integrals of these three signals are 3, 2, and 2, respectively.
Propose structures for compounds 1 and 2, explaining how you reach your conclusion.
The compound G's 1H-NMR spectrum is shown below. Assign the spectrum (feel free to
just use arrows from the chemical structure drawing to the spectrum signals).
.N*
9
8
7
6
5
4
3
PPM
-2
Assign the 13C-NMR spectrum for the compound G above (feel free to just use arrows
from the chemical structure drawing to the spectrum signals).
кадж
180
160
140
120
100
PPM
80
60
40
20
-0
Chapter 13 Solutions
Organic Chemistry (9th Edition)
Ch. 13.5A - In a 300-MHz spectrometer, the protons in...Ch. 13.5B - Prob. 13.2PCh. 13.6 - Determine the number of different kinds of protons...Ch. 13.6 - Prob. 13.4PCh. 13.7 - Draw the integral trace expected for the NMR...Ch. 13.7 - Prob. 13.6PCh. 13.8C - Draw the NMR spectra you would expect for the...Ch. 13.8D - Draw the NMR spectra you expect for the following...Ch. 13.8D - a. Assign protons to the peaks in the NMR spectrum...Ch. 13.8D - Prob. 13.10P
Ch. 13.8D - Two spectra are shown. Propose a structure that...Ch. 13.9 - Prob. 13.12PCh. 13.9 - The spectrum of trans-hex-2-enoic acid follows. a....Ch. 13.9 - Prob. 13.14PCh. 13.9 - Prob. 13.15PCh. 13.10 - Prob. 13.16PCh. 13.10 - If the imaginary replacement of either of two...Ch. 13.10 - Predict the theoretical number of different NMR...Ch. 13.11B - Prob. 13.19PCh. 13.11B - Prob. 13.20PCh. 13.11B - Prob. 13.21PCh. 13.11B - Prob. 13.22PCh. 13.11B - Prob. 13.23PCh. 13.11B - Prob. 13.24PCh. 13.12E - Draw the expected broadband-decoupled 13 C N M R...Ch. 13.12E - a. Show which carbon atoms correspond with which...Ch. 13.12E - Repeat Problem13-25, sketching the...Ch. 13.12F - Prob. 13.28PCh. 13.13 - A bottle of allyl bromide was found to contain a...Ch. 13.13 - A laboratory student was converting cyclohexanol...Ch. 13.14 - Sets of spectra are given for two compounds. For...Ch. 13 - An unknown compound has the molecular formula C 9...Ch. 13 - Prob. 13.34SPCh. 13 - Predict the approximate chemical shifts of the...Ch. 13 - Prob. 13.36SPCh. 13 - Prob. 13.37SPCh. 13 - Prob. 13.38SPCh. 13 - Prob. 13.39SPCh. 13 - Prob. 13.40SPCh. 13 - For each compound shown below. 1. sketch the 13 C...Ch. 13 - Prob. 13.42SPCh. 13 - Prob. 13.43SPCh. 13 - Prob. 13.44SPCh. 13 - Prob. 13.45SPCh. 13 - Prob. 13.46SPCh. 13 - A compound was isolated as a minor constituent in...Ch. 13 - Prob. 13.48SPCh. 13 - The three isomers of dimethylbenzene are commonly...Ch. 13 - a. Draw all six isomers of formula C 4 H 8...Ch. 13 - Prob. 13.51SPCh. 13 - Hexamethylbenzene undergoes free-radical...Ch. 13 - Each of these four structures has molecular...Ch. 13 - Prob. 13.54SPCh. 13 - Phenyl Grignard reagent adds to 2-methylpropanal...Ch. 13 - Prob. 13.56SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 12. H₂C H₂C 1900 4000 CH₂ CH₂ H₂C CH₂ H₂C HSP-47-293 3000 3000 NMR Spectrum ul CH₂ 6 CH3OH CH₂CH3OH CH3CEN Which one of these 17 compounds is represented by this IR and this ¹H NMR spectrum? IR Spectrum тыры H₂C 2009 2000 CH₂ ppm H₂C 4 OH 1300 H₂C. 1500 CH₂ O-CH3 1000 OH H₂C 1000 CH₂ o CH₂arrow_forward4. Match the protons in the molecule with their 'H NMR signals. В нн нн ethyl butyrate (piña colada) H. H Ан H H H НЕ H 4.5 4.0 3.5 3.0 2.5 2.0 1.5 1.0 PPM 0.5 2 2 3 3arrow_forwardIdentify the carbon atoms that give rise to the signals in the 13C NMR spectrum of each compound. a.CH3CH2CH2CH2OH; 13C NMR: 14, 19, 35, and 62 ppm b.(CH3)2CHCHO; 13C NMR: 16, 41, and 205 ppm c.CH2=CHCH(OH)CH3; 13C NMR: 23, 69, 113, and 143 ppmarrow_forward
- Compound 1 has molecular formula C7H16. It shows three signals in the 1H-NMR spectrum, one at 0.85 ppm, one at 1.02 ppm, and one at 1.62 ppm. The relative integrals of these three signals are 6, 1, and 1, respectively. Compound 2 has molecular formula C7H14. It shows three signals in the 1H-NMR spectrum, one at 0.98 ppm, one at 1.36 ppm, and one at 1.55 ppm. The relative integrals of these three signals are 3, 2, and 2, respectively. Propose structures for compounds 1 and 2, explaining how you reach your conclusion.arrow_forward16. A compound with molecular formula CsH,1002 has the following 'H NMR spectrum: Proton NMR 5.0 4.5 4.0 3.5 3.0 2.5 2.0 1.5 1.0 0.5 ppm 모 6.33 Integration Values 19.4 37.9 Determine the number of protons giving rise to each signal.arrow_forwardThe 13C NMR spectra (decoupled 13C 1H spectrum and 3 DEPT spectra) and 1H NMR spectra of the compound in CDC13are shown below. Its molecular formula is C6H1002. Explain why the answer to this spectrum is this molecule -3495.6 1D Carbon DEPT45 ամուր DEPT90 DEPT135 www 200 7.0 1.0 18866 Loave 6.5 -3474.9 3473.2 7.0 180 -3459.3 6.0 elllle ppm 1.0 160 5.5 - 2922.2 2920.5 02162 5.9 wanando 5.0 140 ма 4.5 2906.6 2904.9 2903.2 2901.5 2.0 B 120 4.0 100 f1 (ppm) 3.5 3.0 2091.3 2084.2 2077.1 2069.9 CDCI 3 80 2.5 2.0 60 1.5 6879 933.6 3.0 926.7 924.9 5.8 ppm 4.2 ppm Figure 2: ¹H NMR spectra recorded in CDC13. 3.0 1.9 40 ppm ppm سال I 20 L 0 *** 1.3 ppmarrow_forward
- The DEPT-90 spectrum exhibits 6 in the 0-50 ppm region The DEPT-135 spectrum exhibits x 100 ppm region that is a positive ▾ C6 signal(s) for the CH groups: ▼ 1,2,6 ✓ in the sp2 hybridized region 100-150 C3 and C4 ▼ signal(s) (only the quaternary carbon atoms, signal(s), indicating the presence of a methylene group (CH₂) attached to an oxygen atom, are missing); there is C5 ▼ C1 and C2 ▼ and signal(s) in the 50-arrow_forwardMolecular formula of an unknown compound is, C10H14O. Proton NMR: 1.3 (6H, doublet), 3.2 (1H septet), 4.6 (1H singlet), 2.25 (3H, singlet) 6.7 (1H, singlet), 6.9 (1H, doublet), 7.2 (1H, doublet). What is the name of an unknown compound? 2-isopropyl-5-methylphenol 2-(p-tolyl)propan-2-ol 1-isopropyl-4-methoxybenzene 4-isopropylphenyl)methanolarrow_forwardConstruct a structure of the given 1H NMR data signals.A. C5H12O 0.91 δ (3H, triplet) 1.19 δ (6H, singlet) 1.50 δ (2H, quartet) 2.24 δ (1H, singlet) B. C4H10O 0.90 δ (6H, doublet) 1.76 δ (1H, multiplet) 3.38 δ (2H, doublet) 3.92 δ (1H, singlet) C. C5H10O 1.09 δ (6H, doublet) 2.12 δ (3H, singlet) 2.58 δ (1H, septet)arrow_forward
- Following are the 'H and 13C NMR spectra for each of three isomeric ketones with formula C7H14O. Determine a structure to each pair of spectra and assign each H and C. Carbon spectrum А C,H140 Carbon spectrum В C;H140 CDCI3 200 150 100 50 Proton spectrum CDC13 A C,H140 200 150 100 50 1.96 2.00 2.91 3.0 2.5 2.0 1.5 1.0 0.5 0.0 211.04 -44.79 –17.39 – 13.78 -218.40 - 38.85 –18.55arrow_forwardThe 'H NMR and 13C spectra of a compound with a molecular formula of C6H12O2 are shown below. 1. Name the compound in the textbox below. 2. Draw a possible structure for this compound. 1Η NMR 2H 2H 2H 3HPPM 3H 13C NMR 220 200 100 140 100 120 PPMarrow_forwardAnalyze the high resolution proton NMR spectrum of a compound with the molecular formula C8H16O2 and find out which one. A. 2-ethylhexanoic acid B. 1,4-cyclohexanedimethanol C. ethyl hexanoate D. butyl butyrate E. ethyl 2,2-dimethylpropanoatearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY