
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13, Problem 13.55SP
Phenyl Grignard reagent adds to 2-methylpropanal to give the secondary alcohol shown. The proton NMR of 2-methylpropanal shows the two methyl groups as equivalent (one doublet at δ 1.1), yet the product alcohol, a racemic mixture, shows two different 3H doublets, one at δ 0.75 and one around δ 1.0.
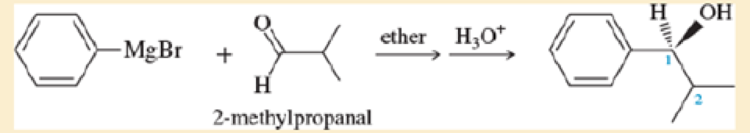
- a. Draw a Newman projection of the product along the C1-C2 axis.
- b. Explain why the two methyl groups have different NMR chemical shifts. What is the term applied to protons such as these?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
9a) The proton bonded to the carbon adjacent to the carbonyl group in the following
compound can be classified into which chemical shift in the ¹H-NMR spectra of aldehydes and
ketones?
R or H
H
- 10.0-12.0 ppm
R
H
-2.0-3.0 ppm
NO₂
N
(1)
9b) Choose the one below that could be resolved into enantiomers.
(1)
alpha hydrogens = ? ppm
NO₂
NO₂
9c) Which of the following is the major product of the reaction below.
`N
H
-5.0-7.0 ppm
(11)
(11)
-0.0-1.0 ppm
HNO3, H₂SO4
H
NO₂
(III)
NO₂
(1) and (II)
(IV)
None of the
options
(IV)
Explain the fact that the C-O stretch in ethers and esters occurs at 1000-1100 cm-1
when the C is sp hybridized, but at 1250 cm- when it is sp2 hybridized.
Determine whether each of the following molecules is a hemiacetal, acetal, or neither and select the appropriate box in the table.
CH₂
CH₂
O
C=0
CH2—0–
HO
CH3
CH₂
0—CH2CH3
CH3
O
OH
X
OH
HỌ–ệo-CH2CH2CH3
Ⓒhemiacetal
O acetal
O neither
Ohemiacetal
O acetal
O neither
Ohemiacetal
O acetal
O neither
Ohemiacetal
O acetal
neither
X
5
Chapter 13 Solutions
Organic Chemistry (9th Edition)
Ch. 13.5A - In a 300-MHz spectrometer, the protons in...Ch. 13.5B - Prob. 13.2PCh. 13.6 - Determine the number of different kinds of protons...Ch. 13.6 - Prob. 13.4PCh. 13.7 - Draw the integral trace expected for the NMR...Ch. 13.7 - Prob. 13.6PCh. 13.8C - Draw the NMR spectra you would expect for the...Ch. 13.8D - Draw the NMR spectra you expect for the following...Ch. 13.8D - a. Assign protons to the peaks in the NMR spectrum...Ch. 13.8D - Prob. 13.10P
Ch. 13.8D - Two spectra are shown. Propose a structure that...Ch. 13.9 - Prob. 13.12PCh. 13.9 - The spectrum of trans-hex-2-enoic acid follows. a....Ch. 13.9 - Prob. 13.14PCh. 13.9 - Prob. 13.15PCh. 13.10 - Prob. 13.16PCh. 13.10 - If the imaginary replacement of either of two...Ch. 13.10 - Predict the theoretical number of different NMR...Ch. 13.11B - Prob. 13.19PCh. 13.11B - Prob. 13.20PCh. 13.11B - Prob. 13.21PCh. 13.11B - Prob. 13.22PCh. 13.11B - Prob. 13.23PCh. 13.11B - Prob. 13.24PCh. 13.12E - Draw the expected broadband-decoupled 13 C N M R...Ch. 13.12E - a. Show which carbon atoms correspond with which...Ch. 13.12E - Repeat Problem13-25, sketching the...Ch. 13.12F - Prob. 13.28PCh. 13.13 - A bottle of allyl bromide was found to contain a...Ch. 13.13 - A laboratory student was converting cyclohexanol...Ch. 13.14 - Sets of spectra are given for two compounds. For...Ch. 13 - An unknown compound has the molecular formula C 9...Ch. 13 - Prob. 13.34SPCh. 13 - Predict the approximate chemical shifts of the...Ch. 13 - Prob. 13.36SPCh. 13 - Prob. 13.37SPCh. 13 - Prob. 13.38SPCh. 13 - Prob. 13.39SPCh. 13 - Prob. 13.40SPCh. 13 - For each compound shown below. 1. sketch the 13 C...Ch. 13 - Prob. 13.42SPCh. 13 - Prob. 13.43SPCh. 13 - Prob. 13.44SPCh. 13 - Prob. 13.45SPCh. 13 - Prob. 13.46SPCh. 13 - A compound was isolated as a minor constituent in...Ch. 13 - Prob. 13.48SPCh. 13 - The three isomers of dimethylbenzene are commonly...Ch. 13 - a. Draw all six isomers of formula C 4 H 8...Ch. 13 - Prob. 13.51SPCh. 13 - Hexamethylbenzene undergoes free-radical...Ch. 13 - Each of these four structures has molecular...Ch. 13 - Prob. 13.54SPCh. 13 - Phenyl Grignard reagent adds to 2-methylpropanal...Ch. 13 - Prob. 13.56SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 6. Name the following compounds and convert the Fisher projection into corresponding structures with wedge-dash notations. сH-CHз НО. -H Н. Br ČH2CH3 CH2CH3 Н—он Br Н ČH,CH3arrow_forwardA sulfonium ion (R3S+) is a stereogenic center if three different alkyl groups are bonded to sulfur because sulfur is surrounded by four different groups, including its lone pair. In assigning an R or S) is a stereogenic center if three different alkyl groups are bonded to sulfur because sulfur is surrounded by four different groups, including its lone pair. In assigning an designation to sulfur, the lone pair is always assigned the lowest priority (4). SAM, S -adenosylmethionine, is a biologically active sulfonium ion that we will learn about in Section 7.16. Locate all the stereogenic centers in SAM, and assign an R,S designation to each center.arrow_forwardDraw the most stable chair conformation of limonene. Label all the nonequivalent protons in the cyclohexene ring with an asterisk (*). The natural product drawn below is eugenol, derived from cloves, nutmeg, bay leaf, and cinnamon. Predict the 8 values for the 1H NMR for each of the protons. H3CO Ha Hb Hc HO Draw a splitting tree for Ha and Hbarrow_forward
- PQ-17. How is this reaction characterized? (A) only regioselective (B) both regioselective and stereospecific (C) only stereospecific (D) neither regioselective nor stereospecific ~ 1) KMnO4. OH, 0°C 2) H₂Oarrow_forwardLabel the following groups as homotopic, enantiotopic or diastereotopic. If they are enantiotopic groups label them pro-R or pro-S The C-O groups HO OH The hydrogens on this CH₂ group The OH groups M+ NHBoc The CH₂Cl groups of the л-allyl intermediate The hydrogens on a particular CH₂Cl grouparrow_forward8. Two structures of Lipitor (a drug used to lower cholesterol) are shown below. (a) Determine the absolute configuration of each indicated stereocenter. Fill in the correct circle. (b) Determine if the two structures are the same compound or stereoisomers. Fill in the correct circle. (a) НО. Carbon a HO O OH Carbon a: OR OS Carbon b H N Carbon b: R OS of H Carbon c: OR OS OH OH Carbon c F Carbon d: R OS OH Carbon d (b) The two structures are: O the same compound O stereoisomersarrow_forward
- (2R,3S)-2-Bromo-3-phenylbutane undergoes an E2 elimination when treated with sodium methoxide. Draw all possible Newman projections for the bond relevant for the elimination reaction, and use those Newman projections to explain the stereochemical outcome of the reaction. Draw the final product and provide its IUPAC name.arrow_forward(−)-Menthol is the most stable stereoisomer of 2-isopropyl-5-methylcyclohexanol and has the R configuration at the hydroxyl-substituted carbon. (a) Draw the preferred conformation of (−)-menthol. (b) (+)-Isomenthol has the same constitution as (−)-menthol. The configurations at C-1 and C-2 of (+)-isomenthol are the opposite of the corresponding chirality centers of (−)-menthol. Write the preferred conformation of (+)-isomenthol.arrow_forwardDevise a synthesis for the following transitions. ul OCH₂CH3 6-7 steps Ph CH₂OCH3arrow_forward
- For the following reaction H+ (2 moles) a) Propose a reaction mechanism and indicate the IUPAC nomenclature of the reactant and product b) Are there chirals in the product? If your answer is yes, draw the possible ethereoisomers and assign their R or S configuration.arrow_forwardCompounds 1 and 2 were prepared, and the difference in their heats of combustion was found to be 17.2 kJ/mol (J. Am. Chem. Soc. 1961, 83, 606-614): H H 1 Shown below are the lowest-energy conformations of compounds 1 and 2. Identify which drawing matches which compound, and identify which compound has the larger heat of combustion. H H H H |||I 2 H 4 H H The first drawing is compound 1, and compound 1 has a larger heat of combustion because it is the less stable compound. O The first drawing is compound 2, and compound 2 has a larger heat of combustion because it is the less stable compound. The first drawing is compound 2, and compound 2 has a larger heat of combustion because it is the more stable compound. The first drawing is compound 1, and compound 1 has a larger heat of combustion because it is the more stable compound.arrow_forwardArrange the following groups in order of decreasing priority that would allow you to determine E/Z, or R/S. b)-H d)-COOH a)-CH3 c) -Br Provide a string of letters (e.g. abcd) as an answer with the highest priority listed first, lowest priority last: CDAB Arrange the following groups in order of decreasing priority that would allow you to determine E/Z, or R/S. a)-OCH3 b)-CH(CH3)2 c) COCH₂ 3 d)-H Provide a string of letters (e.g. abcd) as an answer with the highest priority listed first, lowest priority last:arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY