
Concept explainers
(a)
Interpretation:
Hydrogen atom that ionizes from the picric acid molecule has to be identified.
Concept Introduction:
The most common representation of oxy acid is given by
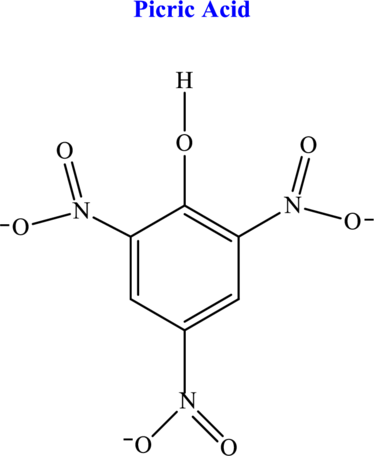
(b)
Interpretation:
Concept Introduction:
The formula to calculate
(c)
Interpretation:
Balanced chemical equation corresponding to decomposition of picric acid has to be written.
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
Whether substitution of
Concept Introduction:
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Chemistry: Principles and Practice
- The pH of a 0.10-M solution of propanoic acid, CH3CH2COOH, a weak organic acid, is measured at equilibrium and found to be 2.93 at 25 °C. Calculate the Ka of propanoic acid.arrow_forwardExplain the difference between a strong acid and a weak acid.arrow_forwardAcids You make a solution by dissolving 0.0010 mol of HCl in enough water to make 1.0 L of solution. a Write the chemical equation for the reaction of HCl(aq) and water. b Without performing calculations, give a rough estimate of the pH of the HCl solution. Justify your answer. c Calculate the H3O+ concentration and the pH of the solution. d Is there any concentration of the base OH present in this solution of HCl(aq)? If so, where did it come from? e If you increase the OH concentration of the solution by adding NaOH, does the H3O+ concentration change? If you think it does, explain why this change occurs and whether the H3O+ concentration increases or decreases. f If you were to measure the pH of 10 drops of the original HCl solution, would you expect it to be different from the pH of the entire sample? Explain. g Explain how two different volumes of your original HCl solution can have the same pH yet contain different moles of H3O+. h If 1.0 L of pure water were added to the HCl solution, would this have any impact on the pH? Explain.arrow_forward
- In a solution prepared by dissolving 0.100 mole of propanoic acid in enough water to make 1.00 L of solution, the pH is observed to be 2.945. The Ka for propanoic acid (HC3H5O2) isarrow_forwardHypochlorous acid, HOCl, is a weak acid having a pKa = 7.46. (a) Show the balanced chemical equation for the hydrolysis of water by OCl- (aq). (b) Compute the value of pKb for aqueous hypochlorite ion. (c) If [OCl-] = 0.050 M, then what is the pH of this aqueous solution? (a) (b) (c)arrow_forward3c.) Suppose it is desired to maintain the pH of a solution at 4.2 using only acetic acid and sodium acetate. Would the concentration of acetic acid or sodium acetate have to be higher in the solution? O sodium acetate O acetic acidarrow_forward
- (a) Given that Ka for acetic acid is 1.8 x 10-5 and that hypochlorous acid is 3.0 x10-8 , which is a stronger acid? (b) Which is the stronger base, the acetate ion or the hypochlorite ion? (c) calculate the Kb values for the CHCOO- and ClO- .arrow_forwardA 0.015 M solution of hydrogen cyanate, HOCN, has a pH of 2.67(a) What is the hydronium ion concentration in the solution?(b) What is the ionisation constant, Ka, for the acid?(c) What is the % ionisation for this solution?arrow_forwardA solution is made by dissolving 44.1 g of Ba(NO₂)₂ in 500.0 mL of water. (a) Does Ba²⁺ have any acidic or basic properties? (b) Does NO₂⁻ have any acidic or basic properties? (c) As NO₂⁻ is a base, write the basic equilibrium equation that exists in solution (d) What is the value of Kb for NO₂⁻? The Ka of HNO₂ is 4.5 × 10⁻⁴. (e) Determine the pH of the solutionarrow_forward
- Sodium hypochlorite solution is also known as bleach. It contains the hypochlorite ion ClO-. (a) Write an equation for the reaction between hypochlorite ion and ammonium ion. Label the acids and bases in the forward and reverse reactions. Identify the two conjugate acid-base pairs. (b) This equilibrium favors the reactants. Which of the acids is stronger and donates protons more readily?arrow_forwardMethylamine, CH3NH2, has Kb= 4.4 x 10–4. (a) Write the ionization equation of methylamine in aqueous solution. (b) Calculate the concentration of OH–and H3O+ in 0.10 M CH3NH2solution. What is the pH ofthe solution?arrow_forward(a) What does the pH scale measure? (b) Define the pH of a substance with hydrogen ion concen- tration of H.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co



