
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 16.67P
Treatment of β-D-glucose with methanol in the presence of an acid catalyst converts it into a mixture of two compounds called methyl glucosides (Section 25.3A).
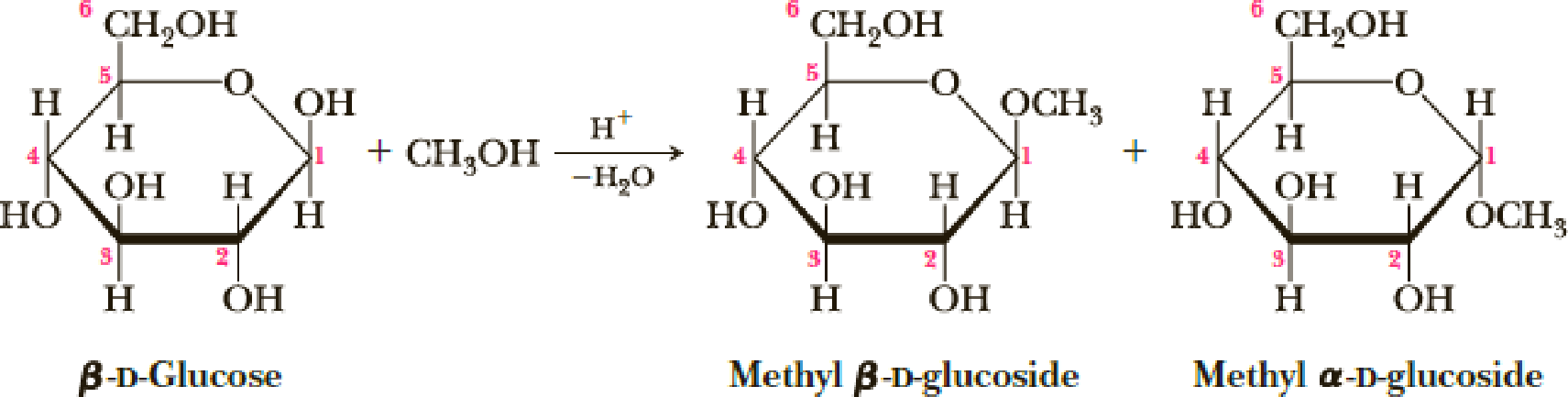
In these representations, the six-membered rings are drawn as planar hexagons.
- (a) Propose a mechanism for this conversion and account for the fact that only the ─OH on carbon 1 is transformed into an ─OCH3 group.
- (b) Draw the more stable chair conformation for each product.
- (c) Which of the two products has the chair conformation of greater stability? Explain.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
(a) Explain how NaBH4 in CH3OH can reduce hemiacetal A to butane-1,4-diol (HOCH2CH2CH2CH2OH). (b) What product is formed when A is treated with Ph3P = CHCH2CH(CH3)2? (c) The drug isotretinoin is formed by reaction of X and Y. What is the structure of isotretinoin? Although isotretinoin (trade name Accutane or Roaccutane) is used for the treatment of severe acne, it is dispensed under strict controls because it also causes birth defects.
Compound X (C4H9Br) reacts by heating with NaOH in H2O to form Y. The
compound Y then undergoes acid catalysed hydration by H2SO4 in 180°C to
form 2-methyl prop-1-ene.
(e) Determine the structure of X and Y.
(f) Predict a MAJOR product when compound Y reacts with H2SO4 in
140°C.
(g) Draw a structural isomer of X. Name the isomer using IUPAC
nomenclature.
(h) Describe a chemical test to distinguish between compound Y and
1-butanol.
Draw the product formed when CH3CH2C=CH is treated with each of the following sets of reagents: (a) H2O, H2SO4, HgSO4; and (b) R2BH, followed by H2O2, HO−.
Chapter 16 Solutions
Organic Chemistry
Ch. 16.1 - Write the IUPAC name for each compound. Specify...Ch. 16.1 - Write structural formulas for all aldehydes with...Ch. 16.1 - Write the IUPAC name for each compound.Ch. 16.5 - Prob. 16.4PCh. 16.6 - Prob. 16.5PCh. 16.7 - Prob. 16.6PCh. 16.7 - Write a mechanism for the acid-catalyzed...Ch. 16.8 - Prob. 16.8PCh. 16.8 - The given mechanism of transamination reaction is...Ch. 16.8 - The given mechanism of transamination reaction is...
Ch. 16.8 - The given mechanism of transamination reaction is...Ch. 16.8 - Prob. DQCh. 16.8 - Prob. EQCh. 16.8 - The given mechanism of transamination reaction is...Ch. 16.9 - Predict the position of the following equilibrium.Ch. 16.9 - Draw a structural formula for the keto form of...Ch. 16.10 - Prob. 16.11PCh. 16.11 - What aldehyde or ketone gives these alcohols upon...Ch. 16.11 - Prob. 16.13PCh. 16 - Prob. 16.14PCh. 16 - Prob. 16.15PCh. 16 - The infrared spectrum of compound A, C6H12O, shows...Ch. 16 - Following are 1H-NMR spectra for compounds B...Ch. 16 - Draw structural formulas for the product formed by...Ch. 16 - Suggest a synthesis for the following alcohols...Ch. 16 - Show how to synthesize the following alcohol using...Ch. 16 - 1-Phenyl-2-butanol is used in perfumery. Show how...Ch. 16 - Prob. 16.22PCh. 16 - Draw structural formulas for (1) the...Ch. 16 - Show how to bring about the following conversions...Ch. 16 - Prob. 16.25PCh. 16 - Wittig reactions with the following -chloroethers...Ch. 16 - Prob. 16.27PCh. 16 - Prob. 16.28PCh. 16 - 5-Hydroxyhexanal forms a six-membered cyclic...Ch. 16 - Prob. 16.30PCh. 16 - Prob. 16.31PCh. 16 - Propose a mechanism to account for the formation...Ch. 16 - Prob. 16.33PCh. 16 - Prob. 16.34PCh. 16 - Show how to bring about the following conversion.Ch. 16 - A primary or secondary alcohol can be protected by...Ch. 16 - Prob. 16.37PCh. 16 - Prob. 16.38PCh. 16 - Prob. 16.39PCh. 16 - Prob. 16.40PCh. 16 - The following molecule belongs to a class of...Ch. 16 - When cis-2-decalone is dissolved in ether...Ch. 16 - Prob. 16.43PCh. 16 - Prob. 16.44PCh. 16 - The following bicyclic ketone has two -carbons and...Ch. 16 - Propose a mechanism for this reaction.Ch. 16 - The base-promoted rearrangement of an -haloketone...Ch. 16 - If the Favorskii rearrangement of...Ch. 16 - (R)-Pulegone, readily available from pennyroyal...Ch. 16 - (R)-Pulegone is converted to (R)-citronellic acid...Ch. 16 - Starting with cyclohexanone, show how to prepare...Ch. 16 - Show how to convert cyclopentanone to these...Ch. 16 - Prob. 16.53PCh. 16 - Prob. 16.54PCh. 16 - Prob. 16.55PCh. 16 - Following is the structural formula of Surfynol, a...Ch. 16 - Prob. 16.57PCh. 16 - Propose a mechanism for this isomerization.Ch. 16 - Starting with acetylene and 1-bromobutane as the...Ch. 16 - Prob. 16.60PCh. 16 - Prob. 16.61PCh. 16 - Prob. 16.62PCh. 16 - Prob. 16.63PCh. 16 - Prob. 16.64PCh. 16 - All rearrangements we have discussed so far have...Ch. 16 - In dilute aqueous base, (R)-glyceraldehyde is...Ch. 16 - Treatment of -D-glucose with methanol in the...Ch. 16 - Treating a Grignard reagent with carbon dioxide...Ch. 16 - Prob. 16.69PCh. 16 - Prob. 16.70PCh. 16 - Prob. 16.71PCh. 16 - Prob. 16.72PCh. 16 - Write the products of the following sequences of...Ch. 16 - Using your reaction roadmaps as a guide, show how...Ch. 16 - Using your reaction roadmaps as a guide, show how...Ch. 16 - Using your reaction roadmaps as a guide, show how...Ch. 16 - Using your reaction roadmaps as a guide, show how...Ch. 16 - Prob. 16.78PCh. 16 - Prob. 16.79PCh. 16 - Prob. 16.80PCh. 16 - Prob. 16.81P
Additional Science Textbook Solutions
Find more solutions based on key concepts
For each of the following 2-dimensional shapes, determine the highest order rotation axis of symmetry.
Inorganic Chemistry
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (14th Edition)
Which of the following solutions has the higher molarity? 10 ppm KI in water or 10,000 ppb KBr in water 0.25 ma...
CHEMISTRY-TEXT
4.1 Write the symbols for the following elements.
a. copper
b. platinum
c. calcium
d. manganese
e. Iron
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Write a Lewis formula for each of the following organic molecules: C2H3Cl (vinyl chloride: starting material fo...
Organic Chemistry - Standalone book
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw and name the seven aldehydes and ketones with the formula C5H10O. Which are chiral?arrow_forwardCiprofloxacin is a member of the fluoroquinolone class of antibiotics.(a) Which of its rings are aromatic?arrow_forward(a) Draw the structure of the hemiacetal formed from one mole of benzaldehyde and one mole of ethanol. (b) Draw the structure of the acetal formed from one mole of benzaldehyde and two moles of ethanol. (c) Draw the structure of 2-methoxy-2-butanol. What compounds could you prepare this from?arrow_forward
- A graduate student was studying enzymatic reductions of cyclohexanones when she encountered some interesting chemistry. When she used an enzyme and NADPH to reduce the following ketone, she was surprised to find that the product was optically active. She carefully repurified the product so that no enzyme, NADPH, or other contaminants were present. Still, the product was optically active. Does the product have any asymmetric carbon atoms or other stereocenters?arrow_forwardA chemist synthesized compound X as a racemic mixture. When the ketone group in X was enzymatically reduced to the corresponding alcohol, a 100% yield was obtained of the product shown below. Choose the statement that best describes this result. ОН enzyme C;H1 `OCH,CH; pH 4.0 C3H1 `OCH,CH3 ОН ÕH X (racemic) (100% yield) One enantiomer of compound X reacts quickly with the enzyme. The other enantiomer of compound X is unreactive, but rapidly equilibrates with the reactive enantiomer under the reaction conditions. Since compound X was racemic, it makes sense that only a single product was obtained. O The product is a meso compound, so either enantiomer of compound X gives the same product. One enantiomer of compound X reacts quickly with the enzyme, while the other enantiomer of compound X remains unchanged.arrow_forwardDraw the organic products formed when each compound is treated with H2, Pd-C. Indicate the three-dimensional structure of all stereoisomers formed.arrow_forward
- Comparing Two Different Methods of Hydration of an Alkene Draw the product formed when CH3CH2CH2CH2CH=CH2 is treated with either (a) H2O, H2SO4; or (b) BH3 followed by H2O2, HO−.arrow_forwardDraw the organic products formed when 2-bromopentan-3-one (CH3CH2COCHBrCH3) is treated with each reagent: (a) Li2CO3, LiBr, DMF; (b) CH3CH2NH2; (c) CH3SHarrow_forward(a) Draw the structure of the hemiacetal formed from one mole of benzaldehyde and one mole of ethanol. (b) Draw the structure of the acetal formed from one mole of benzaldehyde and two moles of ethanol. (c) Draw the structure of 2-methoxy-2-butanol. What compounds could you prepare this from? (d) Draw the structure of 3-methoxyl-2-butanol. What functional groups are present? Is this an acetal, a hemiacetal, or neither? Explain. (e) Identify the functional groups in the molecules shown below. Circle any acetals or hemiacetal, and identify which they are. 0-arrow_forward
- Q2: Write the structure of the product formed in each case (A) C6H₂CH₂CH₂C6H₁ H₂C=CHCCH₂ NaOCH, CH₂OHarrow_forwardDraw the product and indicate the stereochemistry when the given alcohol is treated with each reagent: (a) HBr; (b) PBr3; (c) HCI; (d) SOCI, and pyridine.arrow_forwardName the following aldehydes and ketones: (a) (b) CH2CH2CHO CH3CH2CCHCH3 CH3 Draw structures corresponding to the following names: (b) 4-Chloropentan-2-one (e) 3-Methylbut-3-enalarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License