
You want to create a graph showing the relationship of an ideal gas between pressure (P) and temperature (T). Allow the initial temperature to be 270 kelvin. The range of temperatures to be modeled ranges from 270 to 480 kelvin. Control the scale of the abscissa so the range is shown from 250 to 500 kelvin.
Model two gases:
- Nitrogen (formula, N2; molecular weight, 28 grams per mole), using a 12-liter tank with an initial pressure of 2.5 atmospheres.
- Oxygen (formula, O2; molecular weight, 32 grams per mole), using a 15-liter tank with an initial pressure of 4 atmospheres.
After drawing the graph for nitrogen and oxygen, model a third gas, with information entered by the user. Assume the volume is 12 liters.
Ask the user to enter the name of the gas [Example: Chlorine]
Ask the user to enter the initial pressure in the tank measured at 270 kelvin [Example: 3 atm]
Ask the user to enter the temperature of interest [Example: 400 kelvin]. The user is interested in a temperature in the range of 270 to 480 kelvin.
Output the following information to the Command Window:
At a temperature of TTT kelvin for GGG, the pressure is P.P atm.
where:
- TTT is the temperature of interest entered by the user;
- GGG is the name of the gas; and
- P.P is the pressure at that temperature shown to one decimal place.
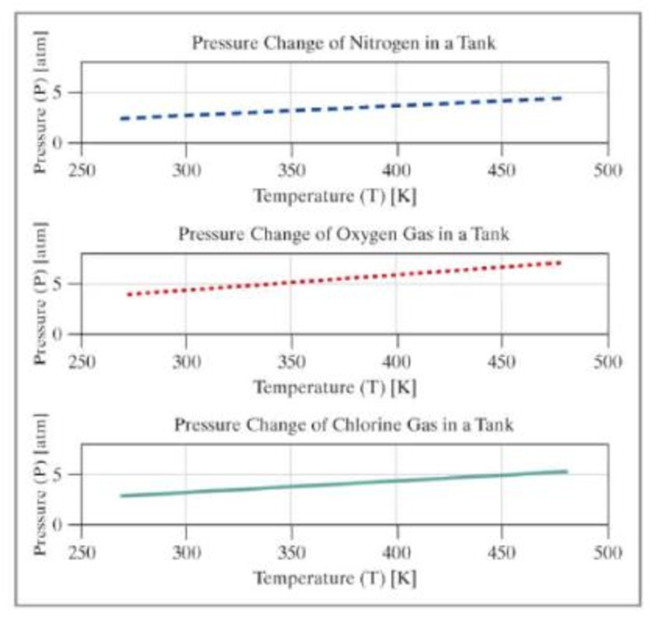
Add the user-entered gas to the graph, showing the pressure over the range of temperatures from 270 to 480 kelvin. In addition to the single graph with three data series, create an additional figure window with a set of subplots showing each data series as an individual subplot.
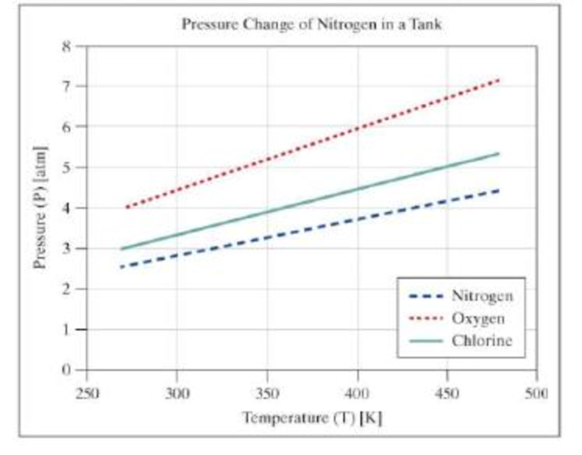
Your output should be similar to the following:
In the Command Window:
Enter the name of the gas: Chlorine
Enter the initial pressure [atm] : 3
Enter the temp of interest [K]: 400
At a temperature of 400 kelvin, chlorine has a pressure of 4.4 atm.
The figures should appear similar to the following. The colors and line types may vary.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Thinking Like an Engineer: An Active Learning Approach (4th Edition)
- The center of mass for a human body can be determined by a segmental method. Using cadavers, it is possible to determine the mass of individual body segments (as a proportion of total body mass) and the center of mass for each segment (often expressed as a distance from one end of the segment). Finding the overall body center of mass can be a complex calculation, involving more than 10 body segments. Below, we will look at a simplified model that uses just six segments: head, trunk, two arms, and two legs. Search y X As a percentage of total body mass, the head is 10%, the two arms are 10%, the trunk is 56%, and the two legs are 24%. The center of mass for each segment is given as an (x,y) coordinate, both units in cm: head = (0, 165), arms = (0, 115), trunk = (0, 95), and legs = (0, 35). Assume the body mass for the individual is 88 kg and their total height is 180 cm. Determine they and y coord 99+ H of massarrow_forwardis a mass hanging by a spring under the influence of gravity. The force due to gravity, Fg, is acting in the negative-y direction. The dynamic variable is y. On the left, the system is shown without spring deflection. On the right, at the beginning of an experiment, the mass is pushed upward (positive-y direction) by an amount y₁. The gravitational constant g, is 9.81 m/s². DO C.D Frontly у Your tasks: No Deflection m k Fg = mg Initial Condition y m k Write down an expression for the total energy If as the sum Write down an expression for the total energy H Fg = mg Figure 3: System schematic for Problem 4. Yi & X Write down, in terms of the variables given, the total potential energy stored in the system when it is held in the initial condition, relative to the system with no deflection. as the sum of potential and kinetic energy in terms of y, y, yi C After the system is released, it will start to move. Write down an expression for the kinetic energy of the system, T, in terms of…arrow_forwardUnder some circumstances, a star can collapse into an extremely dense object made mostly of neutrons and called a neutron star. The density of a neutron star is roughly 10¹4 times as great as that of ordinary solid matter. Suppose we represent the star as a uniform, solid, rigid sphere, both before and after the collapse. The star's initial radius was 6.0x105 km (comparable to our sun); its final radius is 16 km. For related problemsolving tips and strategies, you may want to view a Video Tutor Solution of Anyone can be a ballerina. Part A If the original star rotated once in 29 days, find the angular speed of the neutron star. Express your answer in radians per second. VD| ΑΣΦ W2 = ? rad/sarrow_forward
- I1 Give an example how it is applied in molecular dynamics simulation and Monte carlo simulation? Typical distributions of particles in a volume (e.g. crystal structure for a solid, or distribution of masses and velocities in a “typical” galaxy) - Distributions of particle velocities/energies (e.g. Boltzmann distribution at a fixed temperature) - E.g. for a liquid it is common to start with a solid crystal structure and let the structure “melt” (by setting appropriate velocities corresponding to the liquid phase temperature!) - E.g. to setup a collision of two galaxies, you could try to generate a stable distribution of masses and velocities for a single galaxy first by performing a separate simulation -E.g. A simple model of a phase transition between a low temperature ordered phase (ferromagnet) and high temperature disordered phase (paramagnet) whats the difference in phase space in Molecular dynamics and Monte Carlo simulation?arrow_forwardA population numbers 13,000 organisms initially and grows by 16.2% each year. Suppose P represents population, and t the number of years of growth. An exponential model for the population can be written in the form P = a · b' where P = Preview Get help: Video Videoarrow_forwardI want to run several shocks in my calibrated DSGE model. I want the shock “a” to happen at period 0, shock “b” at period 2 and shock “c” at period 5. Also, I want to run a shock of 1 % to the variable “a”, 2 % to variable “b” and 10 % to variable “c”. How would I write it in the Dynare code? The model is log-linearized around the steady state and all endogenous variables are set at 0 at the steady state.arrow_forward
- The population of a community is known to increase at a rate proportional to the number of people present at time t. If an initial population P, has doubled in 7 years, how long will it take to triple? (Round your answer to one decimal place.) yr How long will it take to quadruple? (Round your answer to one decimal place.) yr еВookarrow_forwardSuppose you're converting a bill of materials for machining feedstock from the British gravitational (U.S. system) of units to International System (SI) units. Your goal is to have all of the material quantities in kilograms. Your bill of materials lists values for required weights of brass rod in pounds. To convert to these values to kilograms you first you divide by gravitational acceleration in feet per second squared to get a mass in slugs, then convert slugs to kilograms using standard conversion factors. Group of answer choices True Falsearrow_forwardAcademic Year 2020 -2021 Q1: A// The thermal conductivity k of a liquid metal is predicted via the empirical equation K = A exp("/r) Where k is in J/(s)(m)(K) and A and B are constants. What are the units of A and B?arrow_forward
- Time left 1:5 An ideal gas has its pressure (1.8) times and mass density (2.2) times increased. If the initial temperature is 265.4 °C, what is the final temperature in °C, and use one number after the decimal (xxx.x)? Answer: NEXT PAGE pe here to search DELLarrow_forwardThermodynamics Answer the following problem with complete solutions. Write legibly A balloon is filled with pure oxygen at 10 ℃. The balloon is released from the bottom of a water-filled testing pool and floats 30 m to the water surface (where the atmospheric pressure is standard). The volume of the balloon is 3,000 cm^3. The temperature is 25 ℃ at the surface of the water, and the water temperature at the bottom of the pool is 10 ℃. What is the volume of the balloon at the surface after it has come into thermal equilibrium with the surroundingsarrow_forwardThe ideal gas law relates the pressure P, volume V, absolute temperature T (Kelvin), and amount of gas n. The law is P = mAT where R is the gas constant. An engineer must design a large natural gas storage tank to be expandable to maintain the pressure constant at 2.2 atmospheres. In December when the temperature is -15°C, the volume of gas in the tank is 28,500 ft°. What will the volume of the same quantity of gas be in July when the temperature is 31°C? (Hint: Use the fact that n, R, and P are constant in this problem. Note also that Kelvin = °C+273.2)arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





