
Interpretation:
Structure of optically inactive triglyceride need to be drawn from the given glycerol, one equivalent of palmitic acid and two equivalents of myristic acid and also to check whether the formed triglyceride will react with molecular hydrogen.
Concept introduction:
Esterification is a process in which the carboxylic acid and an alcohol react to form a ester bond. This can take place in presence of acid or base. The bond is formed due to loss of water molecule when the condensation reaction takes place. Simply to say esterification is the reverse process of hydrolysis.
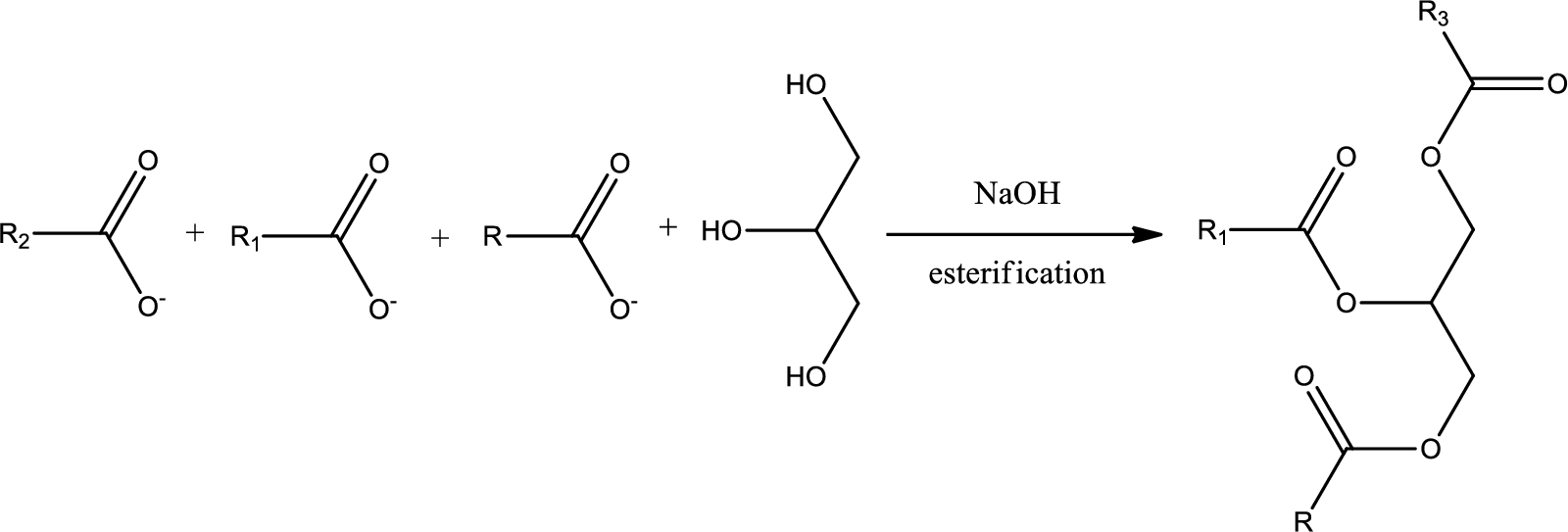
From the above scheme we can find out the starting triglyceride considering the fatty acid and glycerol.
Molecular hydrogen will react with unsaturated compounds to reduce them. If the compound does not have any unsaturation then that compound wont react with molecular hydrogen.
To draw: the structure of optically active triglyceride from the given problem statement.
Want to see the full answer?
Check out a sample textbook solution
Chapter 26 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





