
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 38P
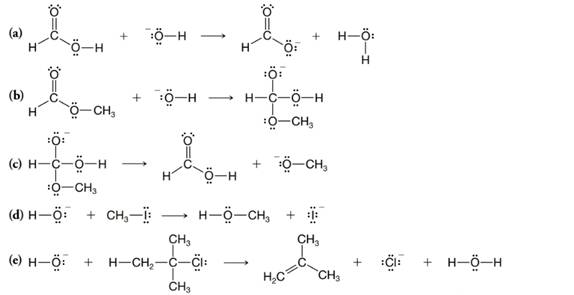
Supply the curved arrows necessary for the following reactions:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which of the following reactions would succeed?
CH3
KMnO4
Acid
? [A]
what is missing to comprete the tho reactian patnuay
below?
HO
1)
OH
2)
Chapter 3 Solutions
Organic Chemistry
Ch. 3 - Prob. 1PPCh. 3 - PRACTICE PROBLEM 3.2
Write equations showing the...Ch. 3 - PRACTICE PROBLEM 3.3 Which of the following are...Ch. 3 - Prob. 4PPCh. 3 - PRACTICE PROBLEM 3.5 Formic acid (HCO2H) has...Ch. 3 - Prob. 6PPCh. 3 - Prob. 7PPCh. 3 - Prob. 8PPCh. 3 - PRACTICE PROBLEM 3.9 Predict the outcome of the...Ch. 3 - Prob. 10PP
Ch. 3 - Prob. 11PPCh. 3 - Prob. 12PPCh. 3 - Prob. 13PPCh. 3 - Prob. 14PPCh. 3 - PRACTICE PROBLEM 3.15 Nitro groups have a large...Ch. 3 - PRACTICE PROBLEM 3.16
Your laboratory instructor...Ch. 3 - Prob. 17PPCh. 3 - Prob. 18PPCh. 3 - Prob. 19PPCh. 3 - What is the conjugate base of each of the...Ch. 3 - List the bases you gave as answers to Problem 3.20...Ch. 3 - 3.22 What is the conjugate acid of each of the...Ch. 3 - List the acids you gave as answers to Problem 3.22...Ch. 3 - Rank the following in order of increasing acidity.Ch. 3 - Without consulting tables, select the stronger...Ch. 3 - Designate the Lewis acid and Lewis base in each of...Ch. 3 - Prob. 27PCh. 3 - Prob. 28PCh. 3 - Write an equation, using the curved-arrow...Ch. 3 - 3.30 What reaction will take place if ethyl...Ch. 3 - 3.31 (a) The of formic acid. What is the? (b)...Ch. 3 - Acid HA has pKa=20; acid HB has pKa=10. (a) Which...Ch. 3 - Prob. 33PCh. 3 - 3.34 (a) Arrange the following compounds in order...Ch. 3 - 3.35 Arrange the following compounds in order of...Ch. 3 - 3.36 Arrange the following in order of increasing...Ch. 3 - Prob. 37PCh. 3 - 3.38 Supply the curved arrows necessary for the...Ch. 3 - Glycine is an amino acid that can be obtained from...Ch. 3 - 3.40 Malonic acid, , is a diprotic acid. The for...Ch. 3 - 3.41 The free-energy change, , for the ionization...Ch. 3 - 3.42 At the enthalpy change, , for the ionization...Ch. 3 - The compound at right has (for obvious reasons)...Ch. 3 - 3.44.
(a) Given the above sequence of...Ch. 3 - Prob. 45PCh. 3 - Prob. 46PCh. 3 - 3.47 As noted in Table 3.1, the of acetone, , is...Ch. 3 - Formamide (HCONH2) has a pKa of approximately 25....Ch. 3 - List all the chemical species likely to be present...Ch. 3 - Prob. 2LGPCh. 3 - Prob. 3LGPCh. 3 - Prob. 4LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
49. The tabulated data show the concentration of AB versus time for this reaction:
Time(s) [AB] (M)
0 0.950
5...
Chemistry: Structure and Properties (2nd Edition)
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
Rank the given solvents in decreasing order of their ability to dissolve each compound. Solutes
Organic Chemistry (9th Edition)
51. Classify each compound as ionic or molecular. If it is ionic, determine whether the metal forms only one ty...
Introductory Chemistry (5th Edition) (Standalone Book)
You are given a sample containing , both with concentrations of 0.020 M. Some of the figures shown on the next ...
Chemistry: The Molecular Nature of Matter
3. Drag out your general chemistry book and solve any four stoichiometry problems dealing with a limiting reage...
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Complete the following equations. If no reaction occurs, write no reaction. a. b. c. d. e.arrow_forwardWrite a sentence or two to explain why the following reaction is not likely to occur:arrow_forwardONH2 (a) Draw the products of the proton transfer reaction shown here. (b) Draw a free energy diagram for this reaction, indicating whether it is endothermic or exothermic. ? + 1arrow_forward
- Predict the product for the following reaction. || excess H₂ Ni, 100 atm, 150°C IV HO Varrow_forwardDoes the equilibrium lie to the left or to the right in the following reaction: H3CO: + H-Br: 2 H;CO-H + H,CO-H + Br:arrow_forwardIn the following given reaction choose the two correct starting material from (a) -(m)arrow_forward
- All reactions are at pH of 0 unless otherwise indicated. Because it has caused confusion in the past, let me be clear. You are asked to draw a poutbaix diagram for the table below.arrow_forwardWhich is most acidic?arrow_forwardIf I have a weak base with a pKb of 6.50 what concentration of [OH-] will be present in a solution at pH of 7? What will the concentration of the conjugate acid be?arrow_forward
- HCl (excess) H₂O₂arrow_forwardList and discuss the basicity of the following compounds.arrow_forwardIn the following reaction in aqueous solution, the acid reactant is and its conjugate base is CH;COOH(aq) + H2O * CH3CO0 (aq) + H30 (aq) A CH3COOH; CH3C00 B CH3COOH; NH4 NH3; CH3CO0 D) NH3; NH4" E CH3COOH; H30*arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY