
Concept explainers
PRACTICE PROBLEM 3.3
Which of the following are potential Lewis acids and which are potential Lewis bases?
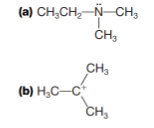
(c)
(d)
(e)
(f)
Trending nowThis is a popular solution!

Chapter 3 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry: Structure and Properties (2nd Edition)
Chemistry: The Molecular Nature of Matter
Chemistry: A Molecular Approach (4th Edition)
Chemistry (7th Edition)
Inorganic Chemistry
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
- (a) (e) Draw the conjugate base for each of the following acids: OH OH (b) (f) H N. (c) NH3 (g) NH4+ (d) H3O+arrow_forward(a) Draw the Lewis structures for at least four species thathave the general formula where X and Y may be the same or different, and n mayhave a value from +1 to -2. (b) Which of the compoundsis likely to be the strongest Brønsted base?arrow_forwardSolve the following Given that pH = 13, 7.5 , 7.4 find: (a) pOH (b) H3O+ (c) OH-arrow_forward
- 2-11 2.19] A solution has an H+ concentration of 10-5 M. (a) What is the pH of this solution? (b) What is the pOH (Assume that the temperature of the solution is 25°C.) Answer: (a) 5 (b) 9 BMarrow_forwardWould CH3NH3C2H3O2 (salt) be neutral, basic, acidic or cannot be determined?arrow_forwardFor the following five reactions: (i) Cl− + AlCl3 → AlCl4−(ii) (CH3)3N + HCl → (CH3)3NHCl(iii) OH− + H3O+ → 2H2O(iv) Co3+ + 6NH3 → Co(NH3)6(v) F− + SiF4 → SiF5− Identify the Lewis acids. Choose from the list below and enter the five letters in order. (e.g. ACEGI, BDFHJ, etc.) A) Cl− B) AlCl3 C) (CH3)3N D) HCl E) OH− F) H3O+ G) Co3+ H) NH3 I) F− J) SiF4arrow_forward
- Consider two acids: HCO2H (formic acid, pKa = 3.8) and pivalic acid [(CH3)3CCO,H, pK = 5.0]. (a) Which acid has the larger K? (b) Which acid is the stronger acid? (c) Which acid forms the stronger conjugate base? (d) When each acid is dissolved in water, for which acid does the equilibrium lie further to the right? %3D The pKa values in Table 2.1 span a large range (-7 to 50). The pK, scale is logarithmic. small difference in pK, translates into a large numerical difference, For example, the diffe between the pK, of NH3 (38) and CH2=CH, (44) is six pKa units. This means that NH, is one million times more acidic than CH,=CH,.arrow_forwardConsider two acids: HCO2H (formic acid, pKa = 3.8) and pivalic acid [(CH3)3CCO2H, pKa = 5.0]. (a) Which acid has the larger Ka? (b) Which acid is the stronger acid? (c) Which acid forms the stronger conjugate base? (d) When each acid is dissolved in water, for which acid does the equilibrium lie further to the right?arrow_forward(a) Calculate [OH-] of a 4.40×10-1 M aqueous solution of triethylamine ((C2H5)3N, Kb = 4.0×10-4). (b)Calculate the pH of the above solution.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





