
Chemistry In Context
9th Edition
ISBN: 9781259638145
Author: Fahlman, Bradley D., Purvis-roberts, Kathleen, Kirk, John S., Bentley, Anne K., Daubenmire, Patrick L., ELLIS, Jamie P., Mury, Michael T., American Chemical Society
Publisher: Mcgraw-hill Education,
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 3.1, Problem 3.6YT
Scientific Practices Energy from the Sun
Examine the distribution of energy from the Sun shown in Figure 3.5.
- a. Which type of
electromagnetic radiation comprises the greatest portion of the energy from the Sun? - b. Which type of radiation is the most intense?
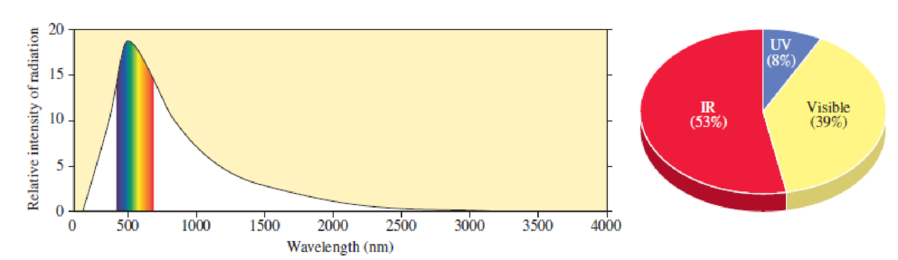
Figure 3.5
Composition of the Sun’s radiation reaching the surface of Earth.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Atom.3 Learning Tracking Tool
Thursday, January 28, 2021
10:49 AM
Due: March 17
Experiment
Claim: What did we figure out?
Answer the experimental question. Summarize
key information.
Key Question
3.1 & 3.2 Flame Test:
Electrons must absorb energy when they
Electrons emit
as they
What determines the
properties of the light
released by different
atoms?
• A larger jump down would release (more/less)
energy and the light would be more (red/violet).
• A smaller jump down would release (more/less)
energy and the light would be more (red/violet)
74.8
Question 6
How much energy is contained in 1 mole of photons with frequency of 8.688 x 1010 s1?
Enter
your answer in joules, as a plain number without scientific notation.
Question 7
Does the Aluminum (Al) ion gain or lose electrons?
a
Lose because it is easier for Aluminum (Al) to get to the Noble Gases losing electrons than gaining electrons
Ob
Lose because Aluminum has a negative charge
Gain because it's on the same level as Argon
Gain because it's easier for Aluminum (Al) to get the Noble Gases gaining electron than losing electrons
Submit
Saved at 8:41 am
Support | Schoology Blog I PRIVACY POLICY I Terms
DELL
Chapter 3 Solutions
Chemistry In Context
Ch. 3.1 - Prob. 3.2YTCh. 3.1 - Using Equation 3.1, answer the following...Ch. 3.1 - Prob. 3.4YTCh. 3.1 - Prob. 3.5YTCh. 3.1 - Scientific Practices Energy from the Sun Examine...Ch. 3.2 - Skill Building Wavelength, Frequency, and Energy...Ch. 3.3 - Skill Building The ABCs of Solar UV a. Arrange...Ch. 3.3 - Prob. 3.9YTCh. 3.4 - Prob. 3.10YTCh. 3.4 - You Decide Exposure Examine the curve shown in...
Ch. 3.4 - Prob. 3.12YTCh. 3.4 - Prob. 3.13YTCh. 3.5 - Prob. 3.14YTCh. 3.5 - Prob. 3.15YTCh. 3.6 - Prob. 3.16YTCh. 3.7 - Draw the Lewis structure for each molecule. a. HBr...Ch. 3.7 - Prob. 3.18YTCh. 3.7 - Prob. 3.19YTCh. 3.7 - You Decide The Ozone Layer Based on the reactions...Ch. 3.8 - Prob. 3.21YTCh. 3.8 - Prob. 3.22YTCh. 3.8 - Prob. 3.23YTCh. 3.8 - Prob. 3.24YTCh. 3.9 - Prob. 3.25YTCh. 3.10 - Prob. 3.26YTCh. 3.10 - Prob. 3.27YTCh. 3.10 - Prob. 3.28YTCh. 3.10 - Prob. 3.29YTCh. 3.11 - Skill Building Particulate Classification An...Ch. 3.11 - Prob. 3.31YTCh. 3.11 - Prob. 3.32YTCh. 3.11 - Prob. 3.33YTCh. 3.11 - Scientific Practices UV Interactions with Matter...Ch. 3 - How does ozone differ from oxygen in its chemical...Ch. 3 - Prob. 2QCh. 3 - Prob. 3QCh. 3 - Prob. 4QCh. 3 - Prob. 5QCh. 3 - a. What is a Dobson unit? b. Does a reading of 320...Ch. 3 - Using the periodic table as a guide, specify the...Ch. 3 - Consider this representation of a periodic table....Ch. 3 - Give the name and symbol for the element with this...Ch. 3 - Prob. 10QCh. 3 - Assuming that the octet rule applies, draw the...Ch. 3 - Prob. 12QCh. 3 - Consider these two waves representing different...Ch. 3 - Use Figure 3.4 to specify the region of the...Ch. 3 - What determines the color of light? Describe the...Ch. 3 - Prob. 16QCh. 3 - Does all light travel at the same speed in a...Ch. 3 - Arrange these types of radiation in order of...Ch. 3 - The microwaves in home microwave ovens have a...Ch. 3 - Ultraviolet radiation is categorized as UVA, UVB,...Ch. 3 - Calculate the wavelength, in nanometers, of the...Ch. 3 - The distance from Earth to the Sun is about 1.50 ...Ch. 3 - Draw Lewis structures for any two different CFCs.Ch. 3 - Prob. 24QCh. 3 - Prob. 25QCh. 3 - Prob. 26QCh. 3 - The following free radicals all play a role in...Ch. 3 - a. How were the original measurements of increases...Ch. 3 - Prob. 29QCh. 3 - The EPA has used the slogan Ozone: Good Up High,...Ch. 3 - Nobel Laureate F. Sherwood Rowland referred to the...Ch. 3 - Prob. 32QCh. 3 - Prob. 33QCh. 3 - Prob. 34QCh. 3 - Prob. 35QCh. 3 - The average length of an OO single bond is 132 pm....Ch. 3 - Prob. 37QCh. 3 - Describe why ozone is more reactive than oxygen...Ch. 3 - Prob. 39QCh. 3 - Prob. 40QCh. 3 - Prob. 41QCh. 3 - All the reports of the damage caused by UV...Ch. 3 - Prob. 43QCh. 3 - Prob. 44QCh. 3 - Prob. 45QCh. 3 - Prob. 46QCh. 3 - Prob. 47QCh. 3 - Development of the stratospheric ozone hole has...Ch. 3 - Prob. 49QCh. 3 - Prob. 50QCh. 3 - Resonance structures can be used to explain the...Ch. 3 - Prob. 52QCh. 3 - Prob. 53QCh. 3 - Prob. 54QCh. 3 - Prob. 55QCh. 3 - Many different types of ozone generators...Ch. 3 - The effect a chemical substance has on the ozone...Ch. 3 - Cooking with an electric stove can have a negative...Ch. 3 - One mechanism that helps break down ozone in the...Ch. 3 - Polar stratospheric clouds (PSCs) play an...Ch. 3 - Prob. 61Q
Additional Science Textbook Solutions
Find more solutions based on key concepts
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
Consider a sample of ideal gas initially in a volume V at temperature T and pressure P. Does the entropy of thi...
General Chemistry: Principles and Modern Applications (11th Edition)
Write a Lewis formula for each of the following organic molecules: C2H3Cl (vinyl chloride: starting material fo...
Organic Chemistry - Standalone book
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (6th Edition)
2. Why shouldn’t you work in a laboratory by yourself?
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
Basic Chemistry (5th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Label the subatomic particles shown in Figure 4.22.arrow_forwardChemistry What is the wavelength of a 27 keV X-ray? a. 4593 Å b. 0.46 Å c. 0.37 Å d. 0.63 Å e. 1240 Åarrow_forwardThis energy diagram shows the allowed energy levels of an electron in a certain atom. (Note: the SI prefix 'zepto' means 10 any SI prefix in the ALEKS Data tab.) -21 You can find the meaning of 1400 - 1200 - 1000 do 800 1 energy (z) -B 600 - 400 -A 200 Use this diagram to complete the table below. What is the energy of the electron in the ground state? What is the energy of the electron in the first excited state? If the electron makes the transition shown by the red arrow, from B to C, will a photon be absorbed or O absorbed Explanation Check © I Privacy Accessibility O 2021 McGraw-Hill Education. All Rights Reserved. Terms of Use FED MacBook Air esc 30 DII DD 4) F1 F2 F3 F4 F5 F7 FB F10 @ %23 24 & 1 1 3 4. 6. 7 8 9. %3D delete Q W R T Yarrow_forward
- PM Thu Sep 16 回9 Q Home Insert Draw Layout Review View bri Regular (B B IU A. 11 1. What is an electron configuration? 2. How is the light emitted by an atom related to its electron configuration? EXPERIMENT 1: THE CHEMISTRY OF FIREWORKS Data Sheet Table 3: Results of Firework Material Ignition Substance Observations Lithium chloride (LiCI) Sodium chloride (NaCI)arrow_forwardWhich of the following scientist’s described a model of the atom that included the energy levels? Bohr Newton Dalton Thomson Galilei A graph with a positive slope means that the variables on the graph are_______ proportional. Jointly Reversely Inverse-square Directly Inverselyarrow_forward2. The speed of light is approximately 3.00×108 m/s. How long does it take light to travel a distance of 10.0 feet? A. 1.01x10-8 s B. 7.14x10-11 s C. 3.00×107 s D. 3.33x10-8 Sarrow_forward
- 29. Cosmic background radiation (CMB) is a form of electromagnetic radiation that fills the entire universe. CMB is considered by some scientists to be evidence of the Big Bang origin of the universe. CMB has a wavelength of approximately 2.0 mm (millimeter). Calculate the frequency of CMB in Hz. Note that Hz = 1/s A) 6.6 × 10-12 B) 1.5 x 10+' C) 1.5 × 10+8 D) 1.5 x 10+5 E) 6.0 × 10+' Hz Hz Hz Hz Hzarrow_forwardQuestion 6:a) Why are most microwave TV dinners packages in plastic rather than aluminum trays? b) The frequency of the radio wave emitted by a cordless telephone is 900 MHz. What is the wavelength of the wave?arrow_forward6. What is the energy of a photon of red light that has a wavelength of 656 nm? a. 2.03 x 10-19 J B. b. 3.03 x 10-19 J c. 4.03 x 10-19 J d. 5.03 x 10-19 Jarrow_forward
- Which type of electromagnetic radiation is the least dangerous to life?A) Microwave B) X-rays C) Ultraviolet D) Radio E) Visiblearrow_forwardAn FM radio station broadcasts electromagnetic radiation at a frequency of 90.2 MHz. The wavelength of this radiation is __________ m a.0.301 b.2.71x10^10 c.3.33 d.2.71x10^16 e.3.33x 10^6arrow_forwardGamma ray radiation has frequencies from 3.0×1019 to 3.0×1024 Hz, whereas the frequency region for microwave radiation is 3.0×108 to 3.0×1011 Hz. We can say that: 1. The speed of gamma ray radiation is _____microwave radiation. A)higher than B)shorter than C)same as The wavelength of gamma ray radiation is _____ microwave radiation. A)Longer than B). Shorter. than C) The same asarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning  Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning


Quantum Numbers, Atomic Orbitals, and Electron Configurations; Author: Professor Dave Explains;https://www.youtube.com/watch?v=Aoi4j8es4gQ;License: Standard YouTube License, CC-BY
QUANTUM MECHANICAL MODEL/Atomic Structure-21E; Author: H to O Chemistry;https://www.youtube.com/watch?v=mYHNUy5hPQE;License: Standard YouTube License, CC-BY