
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 4.8, Problem 4.12P
Under certain conditions, the bromination of cyclohexene follows an unusual rate law:
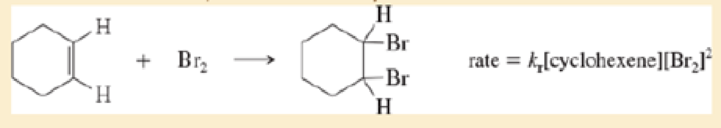
- a. What is the kinetic order with respect to cyclohexene?
- b. What is the kinetic order with respect to bromine?
- c. What is the overall kinetic order?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Nitrogen dioxide (NO₂) concentrations are measured in an air-quality
study and decreases from 10 ppm, to 6 ppm, in 3 min with a particular
light intensity.
a. What is the first-order rate constant for this reaction?
b. What is the half-life of NO₂ during this study?
c.
What would the rate constant need to be changed to in order to
decrease the time required to lower the NO₂ concentration from 10
ppm, to 6 ppm, in 1.5min?
A drug solution decomposes via first-order kinetics with a rate constant, k, of 0.0077 days-1. What is the half-life of the drug in solution?
A.33 days
B. 42 days
C. 90 days
D. 106 days
The dimerization of butadiene:
2 C4H6(g)
C8H12(g)
The following data were obtained:
Time (s)
C16250² 1540 1246 218000682100 |
Determine the order of reaction.
a. cannot be determined.
b. second order
C.
zero order
d. first order
Total number of stereoisomers possible for the following compounds is
a. 6
b. 3
C.
16
d. 4
e.
8
CO
OH
OH
OH
WOH
Chapter 4 Solutions
Organic Chemistry (9th Edition)
Ch. 4.3A - Draw Lewis structures for the following free...Ch. 4.3B - a. Write the propagation steps leading to the...Ch. 4.3C - Prob. 4.3PCh. 4.3C - Prob. 4.4PCh. 4.4 - The following reaction has a value of G =...Ch. 4.4 - Under base-catalyzed conditions two molecules of...Ch. 4.5B - When ethene is mixed with hydrogen in the presence...Ch. 4.5B - For each reaction, estimate whether S for the...Ch. 4.7 - a. Propose a mechanism for the free radical...Ch. 4.7 - a. Using bond-dissociation enthalpies from...
Ch. 4.8 - The reaction of tert-butyl chloride with methanol...Ch. 4.8 - Under certain conditions, the bromination of...Ch. 4.8 - When a small piece of plat num is added to a...Ch. 4.10 - Prob. 4.14PCh. 4.10 - Prob. 4.15PCh. 4.12 - The bromination of methane proceeds through the...Ch. 4.12 - a. Using me BDEs in Table4-2 (page 167 ), compute...Ch. 4.13A - What would be the product ratio in the...Ch. 4.13A - Classify each hydrogen atom in the following...Ch. 4.13B - Use the bond-dissociation enthalpies in Tabte4-2...Ch. 4.13B - Prob. 4.21PCh. 4.13B - Prob. 4.22PCh. 4.14 - a. Compute the heats of reaction for abstraction...Ch. 4.14 - 2,3-Dimethylbutane reacts with bromine in the...Ch. 4.14 - Prob. 4.25PCh. 4.15 - Prob. 4.26PCh. 4.15 - Prob. 4.27PCh. 4.16A - Prob. 4.28PCh. 4.16A - Prob. 4.29PCh. 4.16B - Prob. 4.30PCh. 4.16C - Prob. 4.31PCh. 4.16C - Acetonitrile (CH3C N) is deprotonated by very...Ch. 4.16D - Prob. 4.33PCh. 4 - The following reaction is a common synthesis used...Ch. 4 - Consider the following reaction-energy diagram. a....Ch. 4 - Draw a reaction-energy diagram for a one-step...Ch. 4 - Draw a reaction-energy diagram for a two-step...Ch. 4 - Prob. 4.38SPCh. 4 - Treatment of tert-butyl alcohol with concentrated...Ch. 4 - Label each hydrogen atom in the following...Ch. 4 - Prob. 4.41SPCh. 4 - Prob. 4.42SPCh. 4 - Prob. 4.43SPCh. 4 - Prob. 4.44SPCh. 4 - Prob. 4.45SPCh. 4 - Prob. 4.46SPCh. 4 - For each compound, predict the major product of...Ch. 4 - When exactly 1 mole of methane is mixed with...Ch. 4 - Prob. 4.49SPCh. 4 - Prob. 4.50SPCh. 4 - Prob. 4.51SPCh. 4 - When dichloromethane is treated with strong NaOH,...Ch. 4 - Prob. 4.53SPCh. 4 - When a small amount of iodine is added to a...Ch. 4 - Prob. 4.55SPCh. 4 - When healthy, Earths stratosphere contains a low...Ch. 4 - Prob. 4.57SPCh. 4 - lodination of alkanes using iodine (I2) is usually...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Most reactions occur by a series of steps. The energy profile for a certain reaction that proceeds by a two-step mechanism is On the energy profile, indicate a. the positions of reactants and products. b. the activation energy for the overall reaction. c. E for the reaction. d. Which point on the plot represents the energy of the intermediate in the two-step reaction? e. Which step in the mechanism for this reaction is rate determining, the first or the second step? Explain.arrow_forwardConsider a reaction of the type aA products, in which the rate law is found to he rate = k[A]3 (termolecular reactions are improbable but possible). If the first half-tife of the reaction is found to he 40. s, what is the time for the second half-life? Hint: Using your calculus knowledge, derive the integrated rate law from the differential rate law for a tennolecular reaction: Rate=d[A]dt=k[A]3arrow_forwardWhat is the rate equation for the elementary termolecular reaction A+2B products? For 3A products?arrow_forward
- Three first-order reactions have the following activation energies: (a) Which reaction is the fastest? (b) Which reaction has the largest half-life? (c) Which reaction has the largest rate?arrow_forwardOne experimental procedure that can be used to determine the rate law of a reaction is the method of initial rates. What data are gathered in the method of initial rates, and how are these data manipulated to determine k and the orders of the species in the rate law? Are the units for k. the rate constant, the same for all rate laws? Explain. If a reaction is first order in A, what happens to the rate if [A] is tripled? If the initial rate for a reaction increases by a factor of 16 when [A] is quadrupled, what is the order of n? If a reaction is third order in A and [A] is doubled, what happens to the initial rate? If a reaction is zero order, what effect does [A] have on the initial rate of a reaction?arrow_forwardThe reaction cyclopropane propene occurs on a platinum metal surface at 200 C. (The platinum is a catalyst.) The reaction is first-order in cyclopropane. Indicate how the following quantities change (increase, decrease, or no change) as this reaction progresses, assuming constant temperature. (a) [cyclopropane] (b) [propene] (c) [catalyst] (d) the rate constant, k (e) the order of the reaction (f) the half-life of cyclopropanearrow_forward
- Explain why half-lives are not normally used to describe reactions other than first order.arrow_forwardA certain reaction has the form aAProducts At a particular temperature, concentration versus time data were collected. A plot of 1/ [A] versus Lime (in seconds) gave a straight line with a slope of 6.90 102. What is the differential rate law for this reaction? What is the integrated rate law for this reaction? What is the value of the rate constant for this reaction? If [A]0 for this reaction is 0.100 M, what is the first half-life (in seconds)? If the original concentration (at t = 0) is 0.100 M, what is the second half-life (in seconds)?arrow_forward18. Which of the following statements is FALSE? A. The average rate of a reaction decreases during a reaction. B. It is not possible to determine the rate of a reaction from its balanced equation. C. The rate of zero-order reactions is not dependent on concentration. D. The half-life of a first-order reaction is dependent on the initial concentration of the reactant. E. None of the statements are FALSE.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning  Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Kinetics: Initial Rates and Integrated Rate Laws; Author: Professor Dave Explains;https://www.youtube.com/watch?v=wYqQCojggyM;License: Standard YouTube License, CC-BY