
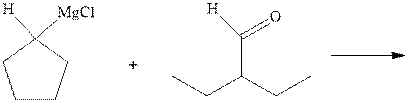
(a)
Interpretation:The mechanism for the major product formation in the indicated reaction should be written.
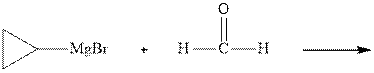
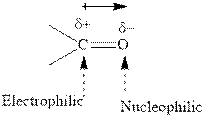
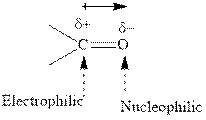
Concept introduction:The carbonyl bond is polar with the partial positive charge on carbon and partial negative charge on oxygen as illustrated below.
Grignard reagents are
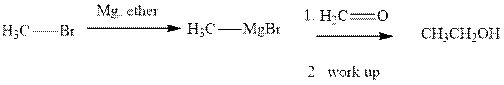
Methyl bromide reacts with magnesium ether to give Grignard reagent. This Grignard methyl magnesium bromide on treatment with formaldehyde gives corresponding alcohol.The mechanistic pathway for the latter reaction is as follows:

The polar carbonyl bond breaks and the polar Grignard reagent attack at electron-deficient carbon. Finally with the hydrolysis and work up the alcohol is formed.
(b)
Interpretation:The mechanism for the major product formation in the indicated reaction should be written.
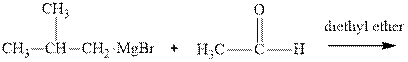
Concept introduction: The carbonyl bond is polar with the partial positive charge on carbon and partial negative charge on oxygen as illustrated below.
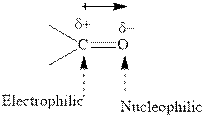
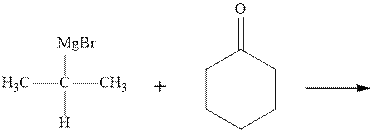
Grignard reagents are alkyl magnesium halides obtained from treatment of haloalkane with magnesium in presence of dried ether conditions. This reagent is useful precursor for quick synthesis of variety of organic compounds For example, the reaction of Grignard reagent with aldehyde or ketone generates alcohol as illustrated below.
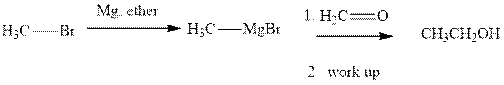
Methyl bromide reacts with magnesium ether to give Grignard reagent. This Grignard methyl magnesium bromide on treatment with formaldehyde gives corresponding alcohol. The mechanistic pathway for the latter reaction is as follows:

The polar carbonyl bond breaks and the polar Grignard reagent attack at electron-deficient carbon. Finally with the hydrolysis and work up the alcohol is formed.
(c)
Interpretation:The mechanism for the major product formation in the indicated reaction should be written.
Concept introduction: The carbonyl bond is polar with the partial positive charge on carbon and partial negative charge on oxygen as illustrated below.
Grignard reagents are alkyl magnesium halides obtained from treatment of haloalkane with magnesium in presence of dried ether conditions. This reagent is useful precursor for quick synthesis of variety of organic compounds For example, the reaction of Grignard reagent with aldehyde or ketone generates alcohol as illustrated below.
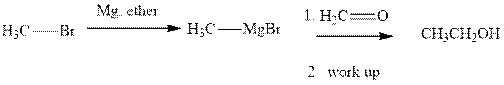
(d)
Interpretation:The mechanism for the major product formation in the indicated reaction should be written.
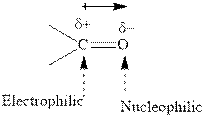
Concept introduction: The carbonyl bond is polar with the partial positive charge on carbon and partial negative charge on oxygen as illustrated below.
Grignard reagents are alkyl magnesium halides obtained from treatment of haloalkane with magnesium in presence of dried ether conditions. This reagent is useful precursor for quick synthesis of variety of organic compounds For example, the reaction of Grignard reagent with aldehyde or ketone generates alcohol as illustrated below.
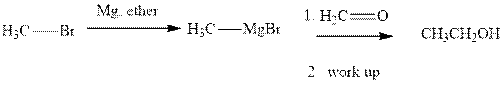
Methyl bromide reacts with magnesium ether to give Grignard reagent. This Grignard methyl magnesium bromide on treatment with formaldehyde gives corresponding alcohol. The mechanistic pathway for the latter reaction is as follows:

The polar carbonyl bond breaks and the polar Grignard reagent attack at electron-deficient carbon. Finally with the hydrolysis and work up the alcohol is formed.
(e)
Interpretation: The mechanism for the major product formation in the indicated reaction should be written.
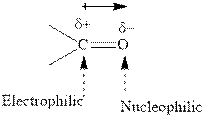
Concept introduction: The carbonyl bond is polar with the partial positive charge on carbon and partial negative charge on oxygen as illustrated below.
Grignard reagents are alkyl magnesium halides obtained from treatment of haloalkane with magnesium in presence of dried ether conditions. This reagent is useful precursor for quick synthesis of variety of organic compounds For example, the reaction of Grignard reagent with aldehyde or ketone generates alcohol as illustrated below.
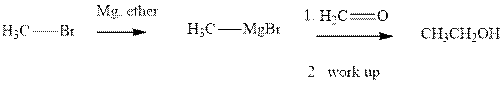
Methyl bromide reacts with magnesium ether to give Grignard reagent. This Grignard methyl magnesium bromide on treatment with formaldehyde gives corresponding alcohol. The mechanistic pathway for the latter reaction is as follows:

The polar carbonyl bond breaks and the polar Grignard reagent attack at electron-deficient carbon. Finally with the hydrolysis and work up the alcohol is formed.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Organic Chemistry: Structure and Function
- Please solve the following reaction mechanisms for various compounds (ketone, aldehyde, alcohol, carboxylic acid, amine) in the presence of diethyl ether (solvent). These reactions occured to test their solubility in ether and all were found soluble.arrow_forwardComplete the following reaction, which may take place in several steps, by supplying all reagents and conditions necessary. Include all important intermediates - mechanisms are not required.arrow_forwardCarry out using the appropriate mechanisms for the reactions given below. Indicate the name of the mechanism used.arrow_forward
- Complete the reaction, show the name of the transformation and give the mechanism Only typed solutionarrow_forwardCan someone show a detailed reaction mechanism for this problem?arrow_forwardA problem often encountered in the oxidation of primary alcohols to acids is that esters are sometimes produced as by-products. For example, oxidation of ethanol yields acetic acid and ethyl acetate: Propose a mechanism to account for the formation of ethyl acetate. Take into account the reversible reaction between aldehydes and alcohols:arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


