
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
4th Edition
ISBN: 9781119776741
Author: Klein
Publisher: WILEY CONS
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 11.3, Problem 6ATS
Interpretation Introduction
Interpretation: A proper synthesis for the following transformation is to be determined.
Concept introduction: The transformation of an alcoholic group to a
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
HOOCH
H CH₂NH2
Tranexamic acid, a drug useful against blood clotting, is prepared commercially from p-methylbenzonitrile. Following is one of the steps in its synthesis, draw the
structure of the product of this step.
COOH
0
CH₂Br
989
་་་
1. excess NH3.
2. H3O+
?
n
?
Previous
Next
C10T03Q1068
Give the major product of the following reaction.
HBr
((CH3)3CO)2
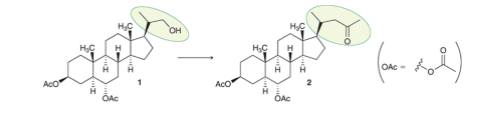
As we will see in Chapter 26, steroids are a class of organic compounds containing a specific tetracyclic skeleton (four rings), as
seen in the compounds shown below. During efforts to synthesize a steroid that was isolated from the Atlantic starfish
(Tetrahedron 2003, 59, 8623-8628), the side chain of compound 1 was converted into the side chain of compound 2, as highlighted
below. Propose a synthesis for this transformation.
Aco
H₂C
O NaOH
O 1) 03; 2) DMS
O NaC=CH
O TSCI, pyridine
H₂C
OAc
OH
Aco
H₂C
H
H₂C
H
ŌAC
2
O
OAc =
This transformation can be accomplished in three steps. Identify the reagent(s) for the first step of the synthesis.
Chapter 11 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
Ch. 11.1 - Prob. 1CCCh. 11.1 - Prob. 2CCCh. 11.2 - Prob. 1LTSCh. 11.2 - Prob. 3PTSCh. 11.3 - Prob. 2LTSCh. 11.3 - Prob. 6ATSCh. 11.4 - Prob. 3LTSCh. 11.4 - Prob. 7PTSCh. 11.4 - Prob. 8ATSCh. 11.5 - Prob. 4LTS
Ch. 11.5 - Propose an efficient synthesis for each of the...Ch. 11.5 - Prob. 10ATSCh. 11 - Prob. 11PPCh. 11 - Prob. 12PPCh. 11 - Prob. 13PPCh. 11 - Prob. 14PPCh. 11 - Prob. 15PPCh. 11 - Prob. 16PPCh. 11 - Prob. 17PPCh. 11 - Prob. 18PPCh. 11 - Prob. 19PPCh. 11 - Prob. 20PPCh. 11 - Prob. 21PPCh. 11 - Prob. 22PPCh. 11 - Prob. 23PPCh. 11 - Prob. 24PPCh. 11 - Prob. 25PPCh. 11 - Prob. 26ASPCh. 11 - Prob. 27ASPCh. 11 - Prob. 28ASPCh. 11 - Prob. 29ASPCh. 11 - Prob. 30ASPCh. 11 - Prob. 31ASPCh. 11 - Prob. 32ASPCh. 11 - Prob. 33IPCh. 11 - Prob. 34IPCh. 11 - Prob. 35IPCh. 11 - Prob. 36IPCh. 11 - Prob. 37IPCh. 11 - Prob. 38IPCh. 11 - Prob. 39IPCh. 11 - Prob. 42CPCh. 11 - The compound Z-3-hexenyl acetate is one of several...Ch. 11 - When a consumer purchases a tomato, smell is one...Ch. 11 - Prob. 45CPCh. 11 - Prob. 46CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- When ethoxybenzene is treated with a mixture of nitric acid and sulfuric acid, two products are obtained, each of which has the molecular formula C8H9NO3. For the mechanism, draw the curved arrows as needed. Include lone pairs and charges in your answer. Do not draw out any hydrogen explicitly in your products. Do not use abbreviations such as Me or Ph.arrow_forwardWhich of the following is an effective way to complete the synthes below. - © LĨNH, 2) H₂O* 1) HNO₂H₂50 2) H₂/Ethanol (can't reduce ring) O DILDA 2) NANO 1) NaOH 2)CHÍNH)arrow_forwardAcyl transfer (nucleophilic substitution at carbonyl) reactions proceed in two stages via a "tetrahedral intermediate." Draw the tetrahedral intermediate as it is first formed in the following reaction. You do not have to consider stereochemistry. Include all valence lone pairs in your answer. Do not include counter-ions, e.g., Na+, I-, in your answer. In cases where there is more than one answer, just draw one.arrow_forward
- Can you please help show how to use the given reactant in order to synthesize this molecule?arrow_forwardprovide the hydrotreating for the organic compoud C9H7N in a more detailed way? regarding which bond will break first and how many steps the reactant will go through to reach the product shown.arrow_forwardAlkenes can be converted into alcohols by acid-catalyze addition of water. Predict the major alcohol product from this alkene below.arrow_forward
- Q17. Compound 1 can undergo an intramolecular reaction to give cyclic product 2. Using curly arrows, show the mechanism of this reaction – note that the mechanism involves more than one elemental step - and draw the structure of 2; chemical formula is provided as guidance. H2N. C5H9NO CH;OH 2arrow_forwardHow do we synthesize compound 2 from benzene?arrow_forwardComplete the mechanism of hydration of alkene below. Please follow curved arrows to predict the resulting structures in each step Step 1 of hydration of 2-methyl-1-propene (Note that the most stable carbocation is formed here) Step 2 of hydration of 2-methyl-1-propene(Note that the most stable carbocation is formed here) Step 3 of hydration of 2-methyl-1-propenearrow_forward
- d) Synthesize the following compound starting from benzene. NO₂arrow_forwardMechanism 2. Provide the complete mechanism for the reactions below. You must include appropriate arrows, intermediates, and formal charges. The ChemDraw template of this document is available on Carmen. OH H2SO4 (cat) THF OHarrow_forwardWhen 5-hexen-3-one is reacted with slightly less H2 than substrate, along with Pt, Pd or Ni metal, the major substrate product will contain the following functional groups: alkene & alcohol. alkene & ketone, since no reaction occurs under these conditions. alkene only. ketone only. alcohol only.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY