
Interpretation:
The Newman projection has to be drawn and the reason has to be explained for the formation of more trans product.
Concept introduction:
Conformations: Rotation about C-C single bonds allows a compound to adopt a variety of possible three-dimensional shapes.
Newman projections: The new conformations of compounds can be drawn and analyzed by Newman projections. A Newman projection visualizes different conformations of Carbon-carbon
The angle between two hydrogens of a Newman projection is called as dihedral angle or torsional angle. This dihedral angle changes as the C-C bond rotates. Two conformations with special attentions are staggered and eclipsed conformation. Staggered conformation is the lowest in energy and the eclipsed conformation is the highest in energy.
For example,

Anti-conformation: The conformation with a dihedral angle of is called anti-conformation.
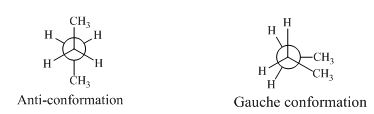
The two methyl groups achieve maximum separation from each other. In other, methyl groups are closer to each other; their electron clouds are repelling each other, causing an increase in energy. This unfavorable interaction is called gauche interaction.
E2 elimination:
Trending nowThis is a popular solution!

Chapter 11 Solutions
Organic Chemistry
- Write Newman projections for all three staggered conformations of (R)-2-bromobutane, viewing down the C2-C3 bond. Upon treatment with strong base (e.g., NAOH), three alkene isomers can form. Illustrate which of the above conformations would be expected to lead to two of these isomers, each with a double bond between C2 and C3. Predict which will be the major product and explain why.arrow_forward(a) When cis-1-bromo-2-methylcyclohexane undergoes an E2 reaction, two products (cycloalkenes) are formed. What are these two cycloalkenes, and which would you expect to be the major product? Write conformational structures showing how each is formed. (b) When rans-1-bromo-2-methylcyclohexane reacts in an E2 reaction, only one cyclo- alkene is formed. What is this product? Write conformational structures showing why it is the only product.arrow_forwardThe ground rules are: you may start with anything that incorporates no more than 5 carbons in your structure per reaction with the exceptions that halobenzenes, phenol, and thiophenol are allowed and work towards this final product. (Z)-1-ethyl-2-(1-phenyl-6-propoxyhept-2-en-1- yl)disulfanearrow_forward
- Reaction of this bicycloalkene with bromine in carbon tetrachloride gives a trans dibro- mide. In both (a) and (b), the bromine atoms are trans to each other. However, only one of these products is formed. CH3 CH3 CH3 Br Br CH,Cl, + Br2 or Br Br (a) (b) Which trans dibromide is formed? How do you account for the fact that it is formed to the exclusion of the other trans dibromide?arrow_forwardDraw the bond-line (skeletal) structure of the compound with molecular formula C₂H₁3Br that gives the following alkene as the exclusive product of E2 elimination. Click and drag to start drawing a structure. C X C Ś c+arrow_forwardWhen it is treated with sodium ethoxide in deuterated ethanol (C2H5OD), cyclopentadiene exchanges all of its hydrogens for deuterium. Draw a structure for the intermediate in this reaction with all protons (no deuterons).arrow_forward
- 1. Predict the elimination products of the following reactions. When two alkenes are possible, predict which one will be the major product. Explain your answers, showing the degree of substitution of each double bond in the products. 2. Which of these reactions are likely to produce both elimination and substitution products? (a) 2-bromopentane +NaOCH3 (b) 3-bromo-3-methylpentane +NaOMe(Me= methyl, CH3) (c) 2-bromo-3-ethylpentane +NaOH (d) cis-1-bromo-2-methylcyclohexane +NaOEt (Et= ethyl, CH2CH3)arrow_forwardCompound A, C₂H16 reacts with 1 molar equivalent(s) of hydrogen on catalytic hydrogenation. A undergoes reaction with ozone, followed by Zn treatment, to give: O CH3 O || CH3CCH₂CCH₂CCH3 CH3 Compound A Propose a structure for A. • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one.arrow_forwardTreatment of propadiene (an allene) with hydrogen bromide produces 2-bromopropene as the major product. This suggests that the more stable carbocation intermediate is produced by the addition of a proton to Br HBr. H2C=C=CH, H3C CH2 a terminal carbon rather than to the central carbon. Propadiene 2-Bromopropene (a) Draw both carbocation intermediates that can be produced by the addition of a proton to the allene. (b) Explain the relative stabilities of those intermediates. Hint: Draw the orbital picture of the intermediates and consider whether the CH, groups in propadiene are in the same plane.arrow_forward
- The alkene shown undergoes bromination. (a) Draw the product(s) of bromination of this compound, including all expected stereoisomers (if any). Use wedge‑and‑dash bonds to designate the stereochemistry at any chirality centers, and make sure to draw an explicit hydrogen if a chirality center has one. (b) Characterize the starting alkene as having the E or Z configuration. (c) characterize the product(s).arrow_forwardThe alkene shown undergoes bromination. H (a) Draw the product(s) of bromination of this compound, including all expected stereoisomers (if any). Use wedge-and-dash bonds to designate the stereochemistry at any chirality centers, and make sure to draw an explicit hydrogen if a chirality center has one. (b) Characterize the starting alkene as having the E or Z configuration. (c) characterize the product(s). (a) H Br₂ Draw the product(s) of bromination. Br H Brarrow_forwardAddition of HBr to allene (CH2=C=CH2) forms 2-bromoprop-1-ene ratherthan 3-bromoprop-1-ene, even though 3-bromoprop-1-ene is formed froman allylic carbocation. Considering the arrangement of orbitals in theallene reactant, explain this result.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
