
Interpretation:
The reason has to be explained for formation nitrile product with S stereochemistry when the reaction of S tosylate with cyanide ion.
Concept introduction:
SN2 reaction:
The alcohol is reaction with acids like hydrochloric acid or hydrobromic acid, the bromine atom attacks back side of the carbon atoms in simultaneous manner and which is bearing alcohol group which yield the corresponding product.
Example:
Alcohol is reaction with tosyl chloride in pyridine which provides retention of configuration of tosylated compound. This tosylated compound is further reaction with sodium methoxide which undergoes again SN2 type of reaction, the methoxide ion attacks the carbon atom through the back side and provides Inverse configuration of methoxy compound. This is shown below,
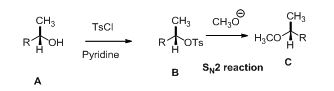
R and S nomenclature:
It is used to assign the molecule using CIP rules.
The CIP rules are as follows:
Select the chiral carbon and assign the numbers according to the decreasing
If the numbering follows clockwise direction then the molecule is termed as R and if it follows anti-clockwise direction then molecule is termed as S.
Given information:
The given compound is shown below,
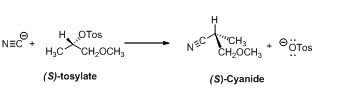
Trending nowThis is a popular solution!

Chapter 11 Solutions
Organic Chemistry
- Predict the product(s) and provide the complete mechanism for each reaction below.arrow_forwardPredict the MAJOR product by proposing a plausible mechanism. Indicate stereochemistry when applicable. NaH Et .arrow_forwardIdentify the following pericyclic reaction; explain the course and stereochemistry of the reaction.arrow_forward
- Predict the major product of the following reaction: 1) Hg(OAc)2, H2O 2) NaBH4 ОН ОН Save for Later (racemic) OH (racemic) ОН ОН ?arrow_forwardThe reaction of cyclohexene with iodobenzene under Heck conditions forms E, a coupling product with the new phenyl group on the allylic carbon, but none of the “expected” coupling product F with the phenyl group bonded directly to the carbon–carbon double bond. Draw a stepwise mechanism that illustrates how E is formed.arrow_forwardorg chem give the major product structure (including stereochemistry if its necessary) of the following reactions you have to put the final product with whats givenarrow_forward
- Predict the the regiochemical outcome (major product) for each of the following reaction, and explain why. (Draw complete resonance structures.) CN ÇO,Et EtO Meoarrow_forwardPredict the product of reaction shown. fuming H2SO, H sO2 sO3 он II IV OII OIV OVarrow_forwardAcid-catalyzed hydrolysis of the following epoxide gives a trans diol. Of the two possible trans diols, only one is formed. How do you account for this stereoselectivity?arrow_forward
- Explain why the following bromoketone forms different bicyclic compounds under different reaction conditions:arrow_forward• Provide the final products P and identify the intermediates A and B. CH3I (C6H5)3P A CoHgLi ether || B C6H5CCH3 Parrow_forwardProvide the products for the reactions, including the appropriate stereochemistry. 1) O3 i) 2) DMS H2 ii) Pdarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

