
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 3.SE, Problem 49AP
Interpretation Introduction
Interpretation:
The structure of malic acid is to be drawn.
Concept introduction:
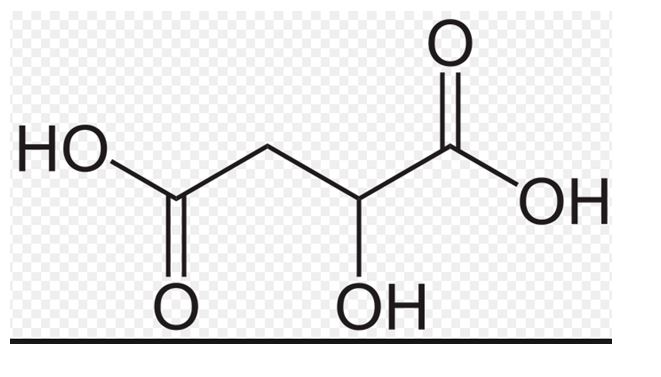
It is a compound present in apple which contributes to the sour taste of it and used as a food additives.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
1. (a) Draw the structures of the eight isomeric pentyl
alcohols, C3H11OH. (b) Name each by the IUPAC system and by the carbinol
system.
(c) Label each as primary, secondary, or tertiary, (d) Which one is isopentyl
alcohol? tert-Pentyl alcohol? (e) Give the structure of a primary, a secondary, and
a tertiary alcohol of the formula C,H13OH.
The foul odor of rancid butter is caused by butyric acid, CH3CH2CH2CO2H.(a) Draw the Lewis structure and determine the oxidation number and hybridization for each carbon atom in the molecule.(b) The esters formed from butyric acid are pleasant-smelling compounds found in fruits and used in perfumes. Draw the Lewis structure for the ester formed from the reaction of butyric acid with 2-propanol.
Draw the structure and name the product formed if the following alcohols are oxidized. Assume an excess of the oxidizing agent is used. If the alcohol is not expected to react with a chemical oxidizing agent, write NR (no reaction).(a) CH3CH2CH2CH2OH(b) 2-butanol(c) 2-methyl-2-propanol(d) 2-methyl-1-propanol
Chapter 3 Solutions
Organic Chemistry
Ch. 3.1 - Prob. 1PCh. 3.1 - Prob. 2PCh. 3.1 - Identify the functional groups in the following...Ch. 3.2 - Draw structures of the five isomers of C6H14.Ch. 3.2 - Propose structures that meet the following...Ch. 3.2 - Prob. 6PCh. 3.3 - Draw the eight 5-carbon alkyl groups (pentyl...Ch. 3.3 - Identify the carbon atoms in the following...Ch. 3.3 - Prob. 9PCh. 3.3 - Prob. 10P
Ch. 3.4 - Give IUPAC names for the following compounds:Ch. 3.4 - Prob. 12PCh. 3.4 - Name the eight 5-carbon alkyl groups you drew in...Ch. 3.4 - Give the IUPAC name for the following hydrocarbon,...Ch. 3.7 - Make a graph of potential energy versus angle of...Ch. 3.7 - Sight along the C2-C1 bond of 2-methylpropane...Ch. 3.7 - Sight along the C2-C3 bond of 2,3-dimethylbutane,...Ch. 3.7 - Draw a Newman projection along the C2-C3 bond of...Ch. 3.SE - Prob. 19VCCh. 3.SE - Prob. 20VCCh. 3.SE - Draw a Newman projection along the C2-C3 bond of...Ch. 3.SE - Prob. 22APCh. 3.SE - Prob. 23APCh. 3.SE - Propose structures for the following: (a) A...Ch. 3.SE - Prob. 25APCh. 3.SE - Draw the structures of the following molecules:...Ch. 3.SE - Draw structures that meet the following...Ch. 3.SE - Prob. 28APCh. 3.SE - In each of the following sets, which structures...Ch. 3.SE - There are seven constitutional isomers with the...Ch. 3.SE - Prob. 31APCh. 3.SE - Draw compounds that contain the following: (a) A...Ch. 3.SE - Prob. 33APCh. 3.SE - Draw and name all monochloro derivatives of...Ch. 3.SE - Draw structures for the following: (a)...Ch. 3.SE - Prob. 36APCh. 3.SE - Draw a compound that: (a) Has nine primary...Ch. 3.SE - Give IUPAC names for the following compounds:Ch. 3.SE - Name the five isomers of C6H14.Ch. 3.SE - Explain why each of the following names is...Ch. 3.SE - Prob. 41APCh. 3.SE - Consider 2-methylbutane (isopentane). Sighting...Ch. 3.SE - What are the relative energies of the three...Ch. 3.SE - Construct a qualitative potential-energy diagram...Ch. 3.SE - Prob. 45APCh. 3.SE - Draw the most stable conformation of pentane,...Ch. 3.SE - Draw the most stable conformation of...Ch. 3.SE - Prob. 48APCh. 3.SE - Prob. 49APCh. 3.SE - Formaldehyde, H2C=O, is known to all biologists...Ch. 3.SE - Prob. 51APCh. 3.SE - Increased substitution around a bond leads to...Ch. 3.SE - Prob. 53APCh. 3.SE - In the next chapter we'll look at...Ch. 3.SE - We’ll see in the next chapter that there are two...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 5.Write the structural formula of the ester that, when hydrolyzed, would yield the following:(a) methanol and propanoic acid(b) 1-octanol and acetic acid (c) ethanol and butanoic acidarrow_forward(a) Draw the four isomers of C₅H₁₀O that can be oxidizedto an aldehyde. (b) Draw the three isomers of C₅H₁₀O that canbe oxidized to a ketone. (c) Draw the isomers of C₅H₁₀O that cannot be easily oxidized to an aldehyde or ketone. (d) Name any isomer that is an alcohol.arrow_forwardAn alcohol is oxidized to a carboxylic acid, and 0.2003 g of the acid is titrated with 45.25 mL of 0.03811 M NaOH.(a) What is the molar mass of the acid? (b) What are the molarmass and molecular formula of the alcohol?arrow_forward
- Draw a structural formula for each alcohol. (a) 2-Methyl-2-propyl-1,3-propanediol (b) 1-Octanol (c) 3,3-Dimethylcyclohexanolarrow_forward5. Give the structural formulae and name the functional groups of the following compounds. (a) 3-chlorobut-1-ene (b) butanedioic acid Name the functional group: (c) propanamide Name the functional group: (d) 3-methylbutanal Name the functional group: Name the functional group:arrow_forwardDraw the condensed structural formula for a carboxylic acid that has the formula C6H12O2, with no substituents.arrow_forward
- Give the structural formulae and name the functional groups of the following compounds. (a) 3-chlorobut-1-ene Name the functional group: (b) butanedioic acid Name the functional group: (c) propanamide Name the functional group: (d) 3-methylbutanal Name the functional group:arrow_forwardWrite a condensed structural formula for a dihydroxy compound with the formula C3H8O2.arrow_forwardWrite two complete, balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures:(a) ethanol reacts with propionic acid(b) benzoic acid, C6H5CO2H, is added to a solution of sodium hydroxidearrow_forward
- The IUPAC name for a carboxylic acid is based on thename of the hydrocarbon with the same number of carbonatoms. The ending -oic is appended, as in ethanoic acid,which is the IUPAC name for acetic acid. Draw the structureof the following acids: (a) methanoic acid, (b) pentanoicacid, (c) 2-chloro-3-methyldecanoic acid.arrow_forwardDraw the hydrogen bonding that takes place between(a) two molecules of ethanol.(b) two molecules of propylamine.(c) a molecule of dimethyl ether and two molecules of water.(d) two molecules of trimethylamine and a molecule of water.arrow_forward1-Propylamine, propan-1-ol, acetic acid, and butane have about the same molar masses. Which would you expect to have the (a) highest boiling point, (b) lowest boiling point, (c) least solubility in water, and (d) least chemical reactivity? Have each member of your group chose a part to answer, andthen discuss with each other why those answers were chosen.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY