
Biochemistry: The Molecular Basis of Life
6th Edition
ISBN: 9780190209896
Author: Trudy McKee, James R. McKee
Publisher: Oxford University Press
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 9, Problem 2Q
Summary Introduction
To review:
Identification of the redox reactions along with their reducing and oxidizing agents from the following reactions:
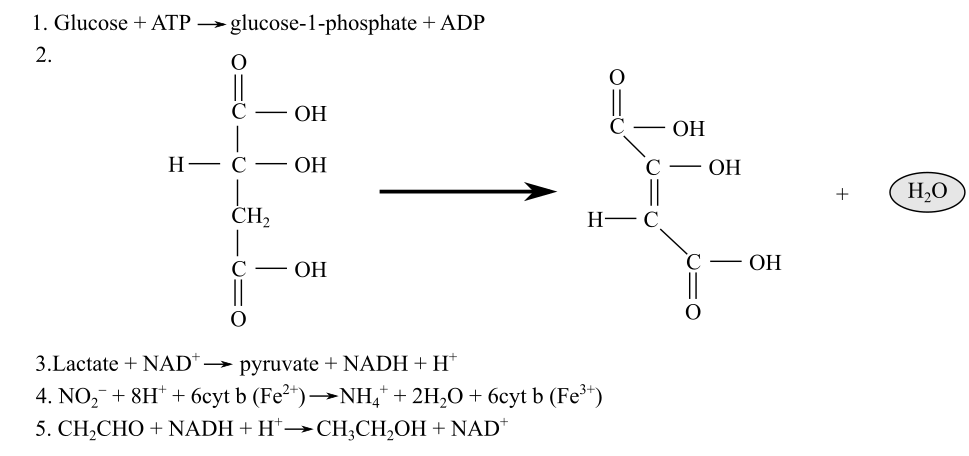
Introduction:
In living organisms, variousredox(
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Complete the balanced equation for the overall reaction by selecting an answer choice in the brackets.
Sucrose + [2 Pi, 4Pi]+[4 ADP, 2 ADP, 4 ATP, 2ATP]+[2 NAD+, 4 NAD+, 6 NAD+]+[H2O, 5 H2O, 3 H2O] --> [2 cirate, 2 oxaloacetate, 2 pyruvate, 2 acetyl-coA]+[4 ADP, 2 ADP, 4 ATP, 2ATP] + [2 NAD+, 4 NAD+, 6 NAD+] + [2H+, 8H+, 6 H+, 4 H+, 10 H+]
Does the commercial process require aerated culture medium—that is, is this a fermentation or an aerobic process?
A. a fermentation process, because A. niger cells must use O2O2 to continuously regenerate NAD+
B. an aerobic process, because A. niger cells must use O2O2 to continuously regenerate NAD+
C. a fermentation process, because A. niger cells cannot use O2O2 to continuously regenerate NAD+
D. an aerobic process, because A. niger cells cannot use O2O2 to continuously regenerate NAD+
A figure of the energy payoff phase of glycolysis is shown below. Look at the
reaction labelled with a 6 (on the far left), which involves the conversion of
glyceraldehyde-3-phosphate
to 1,3-bisphosphoglycerate.
reduced?
HC O
CHOH
CH₂O-P
Glycer-
aldehyde
3-phosphate
(G3P)
2 NADH
2 NAD+ + 2 H+
Triose
phosphate 2 P₁
dehydrogenase
Copyright © 2018 Pearson Canada Inc.
P
CHOH
CH₂0-P
1,3-Bisphospho-
glycerate
1,3- bisphosphoglycerate
NADH
NAD+
2 ADP
GLYCOLYSIS: Energy Payoff Phase
Glyceraldehyde-3-phosphate
2 ATP
Phospho-
glycerokinase
2
Which of the compounds is
C=O
CHOH
H–CO- P
Phospho-
CH₂0-P glyceromutase CH₂OH
8
3-Phospho-
glycerate
C=O
2-Phospho-
glycerate
2. ATP
2 ADP
BYD
Pyruvate
kinase
10
2 H₂O
Enolase
2
C=O
CO-P
CH2
Phosphoenol-
pyruvate (PEP)
CH3
Pyruvate
The reaction pictured is an oxidation-reduction reaction in the citric acid cycle in which the energy-carrier molecule NADH is
generated. Identify which molecule in the reaction will be oxidized and which molecule will be reduced. Place a single answer
choice in each box.
COO-
HO-C-H
H-C-H
COO-
Malate
NAD+ NADH + H+
Oxidized
malate
oxaloacetate
COO-
H-C-H
ī
COO-
Oxaloacetate
Reduced
NADH
NAD+
Chapter 9 Solutions
Biochemistry: The Molecular Basis of Life
Ch. 9 - Prob. 1QCh. 9 - Prob. 2QCh. 9 - Prob. 3QCh. 9 - Prob. 4QCh. 9 - Prob. 5QCh. 9 - Prob. 6QCh. 9 - Prob. 7QCh. 9 - Prob. 1RQCh. 9 - Prob. 2RQCh. 9 - Prob. 3RQ
Ch. 9 - Prob. 4RQCh. 9 - Prob. 5RQCh. 9 - Prob. 6RQCh. 9 - Prob. 7RQCh. 9 - Prob. 8RQCh. 9 - Prob. 9RQCh. 9 - Prob. 10RQCh. 9 - Prob. 11RQCh. 9 - Prob. 12RQCh. 9 - Prob. 13RQCh. 9 - Prob. 14RQCh. 9 - Prob. 15RQCh. 9 - Prob. 16RQCh. 9 - Prob. 17RQCh. 9 - Prob. 18RQCh. 9 - Prob. 19RQCh. 9 - Prob. 20RQCh. 9 - Prob. 21RQCh. 9 - Prob. 22RQCh. 9 - Prob. 23RQCh. 9 - Prob. 24RQCh. 9 - Prob. 25RQCh. 9 - Prob. 26RQCh. 9 - Prob. 27RQCh. 9 - Prob. 28FBCh. 9 - Prob. 29FBCh. 9 - Prob. 30FBCh. 9 - Prob. 31FBCh. 9 - Prob. 32FBCh. 9 - Prob. 33FBCh. 9 - Prob. 34FBCh. 9 - Prob. 35FBCh. 9 - Prob. 36FBCh. 9 - Prob. 37FBCh. 9 - Prob. 38SACh. 9 - Prob. 39SACh. 9 - Prob. 40SACh. 9 - Prob. 41SACh. 9 - Prob. 42SACh. 9 - Prob. 43TQCh. 9 - Prob. 44TQCh. 9 - Prob. 45TQCh. 9 - Prob. 46TQCh. 9 - Prob. 47TQCh. 9 - Prob. 48TQCh. 9 - Prob. 49TQCh. 9 - Prob. 50TQCh. 9 - Prob. 51TQCh. 9 - Prob. 52TQCh. 9 - Prob. 53TQCh. 9 - Prob. 54TQCh. 9 - Prob. 55TQCh. 9 - Prob. 56TQCh. 9 - Prob. 57TQCh. 9 - Prob. 58TQCh. 9 - Prob. 59TQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Complete the balanced equation for the overall reaction. Select answer choice in between brackets. Sucrose + [2 Pi, 4Pi]+[4 ADP, 2 ADP, 4 ATP, 2ATP]+[2 NAD+, 4 NAD+, 6 NAD+]+[H2O, 5 H2O, 3 H2O] --> [2 cirate, 2 oxaloacetate, 2 pyruvate, 2 acetyl-coA]+[4 ADP, 2 ADP, 4 ATP, 2ATP] + [2 NAD+, 4 NAD+, 6 NAD+] + [2H+, 8H+, 6 H+, 4 H+, 10 H+] Does the commercial process require aerated culture medium—that is, is this a fermentation or an aerobic process? A. a fermentation process, because A. niger cells must use O2O2 to continuously regenerate NAD+ B. an aerobic process, because A. niger cells must use O2O2 to continuously regenerate NAD+ C. a fermentation process, because A. niger cells cannot use O2O2 to continuously regenerate NAD+ D. an aerobic process, because A. niger cells cannot use O2O2 to continuously regenerate NAD+arrow_forwardBalance the following net reaction that occurs during oxidative phosphorylation. Use the smallest whole integers possible for the reaction stoichiometry. NADH+ FADH₂ + ADP+ 0₂+H+PO, ¹ NAD+FAD+ ATP+ +H₂Oarrow_forwardWhich of the compounds shown below exhibit a high negative free energy of hydrolysis equal to or larger than the free energy of hydrolysis for the phosphoanhydride of ATP? (Choose all that apply) В Α HPΗ Ho HO HO H2N-CH-C CH2 H2N-CH-C CH2 CH2 H2N-CH-Ö CH2 C CH2 D E O A O B O E O=p-00 O=p-00 O=-00 O=0-00 O=L-00arrow_forward
- Under standard conditions, will the following reaction proceed spontaneously as written? Fumarate + NADH + H+⇌ succinate + NAD+arrow_forwardConsider the reaction: malate + NAD+ → oxaloacetate + NADH + H+. Calculate ΔE°’ for the reaction.arrow_forwardConsider the following reaction of the glycolysis pathway. Which statement is NOT true regarding this reaction? P + C-H NADH + H+ NAD+ C-0-P H-C-OH H-C-OH Glyceraldehyde- CH₂0-P 3-phosphate dehydrogenase CH₂0-P 1,3-Bisphosphoglycerate Glyceraldehyde-3- phosphate O A one-carbon compound (1C) is converted into a two-carbon compound (2C). O It requires a coenzyme. O It is a redox reaction catalyzed by the enzyme glyceraldehyde-3-phosphate dehydrogenase. O It is a phosphorylation reaction.arrow_forward
- Example: Oxidation of ethanol by NAD+ in the presence catalyzed by alcohol dehydrogenase Calculate the standard free energy change for the reaction below: Ethanol + NAD* 2 acetaldehydye + NADH + H*arrow_forwardIndicate the general type of enzyme that mediates each glycolysis reaction depicted below. (e.g. Transferase, Oxidoreductase, Kinase, Hydrolase, Lyase, or Isomerase) iii. O H Glyceraldehyde-3-phosphate → 1,3-Bisphosphoglycerate H-C-OH + NAD CH₂O-P + P₁ H-O- -C-H Glucose →→ Glucose-6-phosphate H b 0 OH HO OH H-C- 0 -H HO- H ОН H-C-OH + NADH +H+ CH₂O P P ATP ADP 1 H (P-O- -C- H H HO OH 2-Phosphoglycerate → Phosphoenolpyruvate 0 OH H ОН -O~(P) + HOHarrow_forwardWhen grown anaerobically on glucose, yeast (S. cerevisiae) converts pyruvate to acetaldehyde, then reduces acetaldehyde to Pethanol using electrons from NADH. Write the chemical equation for the reaction that reduces acetaldehyde (CH3CHO) to ethanol (CH3CH2OH). The table provides the standard reduction potential, E', of the relevant half-reactions. Half-reaction Acetaldehyde + 2 H+ + 2e¯ → ethanol NAD+ + 2H+ + 2e¯ → NADH + H+ E'° (V) -.197 -.320 Calculate the equilibrium constant, K'eq, at 25.0 °C for the reaction that reduces acetaldehyde to ethanol. K'e ×10 = eqarrow_forward
- When grown anaerobically on glucose, yeast (S. cerevisiae) converts pyruvate to acetaldehyde, then reduces acetaldehyde to ethanol using electrons from NADH. Write the equation for the second reaction, and calculate its equilibrium constant at 25 °Carrow_forwardU Select all of the following half reactions that require energy (work) to proceed as written. NAD+ + 2HNADH + H+ glucose + inorganic phosphate glucose-6-phosphate + H₂O › DETAILS O=d-0 OH NADH+H NAD+ + 2H OH o-p-o-C-CH3 + H2O -0-8-CH3 PREVIOUS ANSWERS ATP+ H₂O ADP + P₁ H قمر H (not balanced) (not balanced) O 0-4-OH + HO-CH₂ 0 xarrow_forwardNAD+ coenzyme is required for the reaction of ethanol in the liver to ethanal and NADH. Write down the metabolic reaction below:arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON

Biochemistry
Biochemistry
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:9781464126116
Author:David L. Nelson, Michael M. Cox
Publisher:W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul...
Biochemistry
ISBN:9781118918401
Author:Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:WILEY

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Fundamentals of General, Organic, and Biological ...
Biochemistry
ISBN:9780134015187
Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:PEARSON
Anaerobic Respiration; Author: Bozeman Science;https://www.youtube.com/watch?v=cDC29iBxb3w;License: Standard YouTube License, CC-BY